In the simplest terms, immunotherapy is a type of cancer treatment that uses your own immune system to fight cancer. It’s a way of boosting or redirecting your body’s natural defenses to better find and destroy cancer cells.
The Fourth Pillar of Cancer Care
For a long time, cancer treatment stood on three main pillars: surgery, radiation, and chemotherapy. While these are often powerful tools, they can sometimes harm healthy cells in the process of attacking the cancer, leading to tough side effects. Immunotherapy has now firmly established itself as the “fourth pillar,” marking a major evolution in how we think about treating cancer.
Instead of hitting the tumor directly with external forces like chemicals or radiation, immunotherapy turns the fight inward. Think of your immune system as a highly trained security team patrolling your body. Cancer cells are masters of disguise; they’ve learned tricks to hide from this team or even switch off the alarms that would normally alert them.
Immunotherapy essentially gives your internal security team a new set of orders and better gear. It helps them see through the cancer’s disguise and reawakens the sleeping guards, pointing them directly at the threat they previously missed.
A Long History Behind a “New” Idea
While immunotherapy feels like a cutting-edge breakthrough, the idea has been simmering for over a century. It all started back in 1891, when a surgeon named Dr. William Coley tried injecting bacteria into tumors to kickstart an immune response. This was the seed that would eventually grow into modern immunotherapy.
A huge leap forward came in 1986 when the FDA approved interferon-α, the first modern immunotherapy for an advanced cancer. This proved that boosting the body’s own immune signals could actually fight back against malignancies. These early steps paved the way for the game-changing checkpoint inhibitors of the 2010s, which led to Science magazine naming cancer immunotherapy its ‘Breakthrough of the Year’ in 2013. You can dig into a great timeline of these milestones and learn more about the evolution of immunotherapy treatment on PMC NCBI.
What Makes Immunotherapy So Different?
The real magic of this approach lies in its potential to create a lasting defense. When you stop chemotherapy, its effects stop too. Immunotherapy is different because it can create an “immune memory.” This means that long after treatment is over, your immune system might remember what the cancer cells look like, ready to hunt them down if they ever try to come back.
This unique way of working offers some incredible advantages:
- Precision Targeting: It specifically trains the immune system to go after cancer cells, which often spares healthy tissues from the collateral damage seen with other treatments.
- Long-Lasting Results: For some people, the effects can last for years, putting them into a long-term remission even after they’ve finished their therapy.
- Wide-Ranging Use: It’s proven effective against a whole host of cancers, from melanoma and lung cancer to certain kidney and bladder cancers.
This treatment leverages the patient’s own immune system to fight cancer. It activates and pushes it to find the tumors and kill them. You can use different tools—T cells, vaccines, targeted drugs—but they all use the patient’s immune cells to fight their tumors.
This fundamental shift—working with the body instead of just acting on it—is precisely why immunotherapy is seen as such a monumental step forward in oncology.
How Immunotherapy Teaches Your Body to Fight Cancer
To really get what immunotherapy for cancer is, you have to think about how it cleverly re-educates your body’s own internal security team. Picture your immune cells—especially your T-cells—as highly trained guards constantly patrolling your body. Their main job is to find and take out any threats, like viruses, bacteria, or even your own damaged cells.
But how do they avoid attacking your healthy tissues? They use a sophisticated system of checks and balances, almost like a secret handshake. When a T-cell bumps into a healthy cell, they swap a specific molecular signal. This “handshake” basically tells the T-cell, “I’m one of the good guys, move along.”
This system is called an immune checkpoint, and it’s absolutely critical for preventing autoimmune diseases where the body goes haywire and attacks itself. It’s a natural brake that keeps your powerful immune response from going rogue.
The Cancer Cell’s Clever Disguise
Here’s the problem: cancer cells are dangerously smart. Over time, some of them figure out how to exploit this very system. They learn to put that same “friendly” signal protein on their surface, effectively learning the secret handshake.
So, when a T-cell guard comes along, ready to attack, the cancer cell flashes this false signal. The T-cell is completely fooled. It gets the “I’m friendly” message, hits its brakes, and leaves the dangerous cell to grow and multiply in plain sight.
“Immunotherapy helps the immune system see where the tumor is in the body. The immune system can see a single cancer cell. That’s powerful.”
This molecular camouflage is one of the biggest reasons cancer can get a foothold, even with our body’s defenses on high alert. The security system is there, but the intruders have found a way to blend in perfectly. And that’s exactly where a major type of immunotherapy, called checkpoint inhibitors, comes into play.
Uncloaking the Enemy with Checkpoint Inhibitors
Checkpoint inhibitor drugs are engineered to block this deceptive handshake. They don’t attack cancer cells directly. Instead, they act like a shield, getting in between the cancer cell and the T-cell so they can’t make that “friendly” connection.
Think of it playing out like this:
- The Deceptive Handshake: A cancer cell presents a protein (like PD-L1) to a T-cell’s receptor (like PD-1).
- The Brakes Are Hit: This connection tells the T-cell to back off, and the cancer is left alone.
- Immunotherapy Steps In: A checkpoint inhibitor drug physically blocks either the protein on the cancer cell or the receptor on the T-cell.
- The Disguise Fails: With the handshake blocked, the T-cell never gets the “stand down” signal. It finally recognizes the cancer cell for the threat it is.
- The Attack Begins: The brakes on the T-cell are released, and it launches an attack, destroying the now-unmasked cancer cell.
This whole approach is so powerful because it doesn’t introduce a foreign poison into your body; it just takes down the shield that cancer built to protect itself. By doing that, it unleashes the full, natural force of your immune system to do the job it was always designed to do. Understanding these intricate cellular signals often requires complex computer analysis, and if that field piques your interest, you can learn more about what is bioinformatics in our detailed guide. This process of unmasking cancer is the core principle behind some of the most successful immunotherapy treatments we have today.
Exploring the Main Types of Cancer Immunotherapy
Immunotherapy isn’t a single treatment. Think of it more like a toolbox filled with different, highly specialized tools, each designed to help your immune system fight cancer in its own unique way. An oncologist’s job is to pick the right tool—or combination of tools—for a specific cancer and a specific person.
You wouldn’t use a hammer to turn a screw. In the same way, each type of immunotherapy has a different job to do, a different mechanism for getting it done, and a different set of targets it’s best suited for. Let’s meet the major players on this incredible team.
This diagram helps visualize how immunotherapy works at a high level. It essentially steps in to block the “secret handshake” that cancer cells use to trick immune cells into leaving them alone.
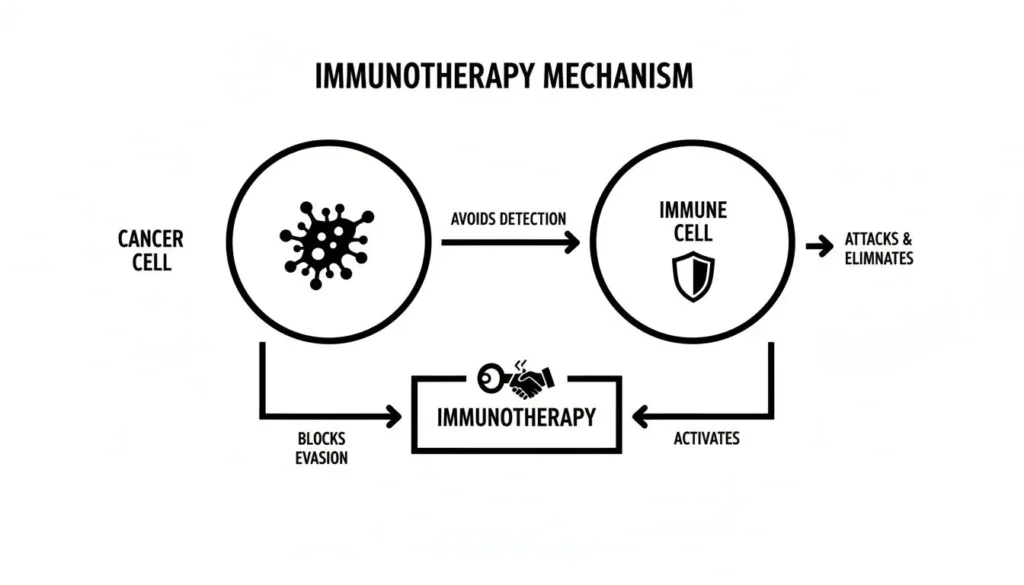
By interrupting this signal, the treatment pulls the mask off the cancer cells, letting your body’s natural defenses finally see them as a threat and launch an attack.
A Quick Look at Different Immunotherapy Approaches
To give you a bird’s-eye view, this table breaks down the main strategies. It’s a simplified snapshot, but it helps show the distinct role each therapy plays in the fight against cancer.
| Immunotherapy Type | How It Works (Simple Analogy) | Commonly Used For |
|---|---|---|
| Checkpoint Inhibitors | “Releasing the brakes” on the immune system so it can attack freely. | Melanoma, lung cancer, kidney cancer, bladder cancer |
| Monoclonal Antibodies | “Guided missiles” that flag or block specific targets on cancer cells. | Breast cancer, colorectal cancer, lymphomas |
| CAR-T Cell Therapy | “Super-soldier training” for your T-cells to hunt down cancer. | Leukemias, lymphomas, multiple myeloma |
| Cancer Vaccines | “Wanted posters” that teach the immune system what cancer cells look like. | Prostate cancer, melanoma (mostly in trials) |
| Oncolytic Viruses | “Trojan horses” that infect and destroy cancer cells from the inside. | Melanoma, other solid tumors (mostly in trials) |
| Cytokines | “System-wide alerts” that boost overall immune activity. | Kidney cancer, melanoma (less common now) |
Each of these approaches represents a different way to weaponize the body’s own defenses. Now, let’s dive into how some of the most common ones work in more detail.
Immune Checkpoint Inhibitors: Releasing the Brakes
As we covered earlier, your immune system has natural “brakes,” called checkpoints, to keep it from going overboard and attacking healthy tissue. The trouble is, many cancers have learned how to slam on those brakes to protect themselves.
Immune checkpoint inhibitors are drugs that do exactly what their name suggests: they block those checkpoints. They essentially cut the brake lines, freeing up your immune cells—especially T-cells—to attack the cancer with full force.
These drugs have been game-changers for a growing list of cancers, including:
- Melanoma
- Non-small cell lung cancer
- Kidney cancer
- Bladder cancer
Monoclonal Antibodies: The Smart Missiles
Monoclonal antibodies are precision-engineered proteins that act like guided missiles. In the lab, scientists design them to hunt for and lock onto very specific proteins (called antigens) that are found on the surface of cancer cells.
Once they’ve found their target, they can work in a few different ways. Some simply “flag” the cancer cell, making it an obvious target for other immune cells to find and destroy. Others might directly interfere with proteins that the cancer cell needs to grow or spread. It’s this targeted approach that makes them so effective.
CAR-T Cell Therapy: The Living Drug
One of the most exciting frontiers in cancer treatment is CAR-T cell therapy. This is a highly personalized treatment that turns a patient’s own immune cells into a powerful, cancer-fighting army.
Here’s how it works: doctors take a sample of your T-cells, send them to a lab where they are genetically engineered, and then infuse them back into your body. The engineering part gives the T-cells a new “guidance system”—a Chimeric Antigen Receptor, or CAR—that allows them to recognize and kill your specific cancer cells. You can think of it as giving your T-cell soldiers a set of night-vision goggles that only see cancer.
This “living drug” can then multiply inside your body, creating a long-lasting surveillance team on the lookout for cancer. It has produced incredible results for patients with certain blood cancers, like leukemia and lymphoma. Because it involves modifying a patient’s own genetic material, it shares some common ground with gene therapy. You can read more about that in our guide explaining what gene therapy is and how it works.
Cancer Vaccines: The Wanted Posters
Most of us think of vaccines as something you get to prevent a disease. But cancer treatment vaccines are different; they’re designed to help your body fight a cancer that’s already there.
A cancer vaccine works by showing your immune system a piece of the cancer—an antigen—to train it to recognize the threat. It’s like putting up a “wanted poster” all over your body with a mugshot of the cancer cell. This teaches your immune system what to look for, so it can mount a specific, targeted attack. While many are still being tested in clinical trials, they represent a very promising future direction.
Other Important Immunotherapy Types
Beyond the big names, a few other strategies play important roles in the immunotherapy landscape.
- Cytokines: These are messenger proteins that act as a general “red alert” for your immune system. They don’t target cancer directly but rather boost the overall activity of all your immune cells, putting them on high notice.
- Oncolytic Viruses: This approach uses viruses—either naturally occurring or modified in a lab—that are programmed to selectively infect and kill cancer cells. When the virus makes a cancer cell burst open, it spills cancer antigens everywhere, which can trigger an even broader immune response against any remaining tumor cells.
Weighing the Pros and Cons: Benefits vs. Potential Side Effects
When you’re considering immunotherapy, it’s a conversation about incredible promise on one hand and potential challenges on the other. This isn’t like other cancer treatments, so the outcomes—and what you need to watch for—are totally unique. The whole idea is to unleash your own body’s defenses, but that powerful process follows its own set of rules.

Here’s the key difference from something like chemotherapy: chemo stops working the moment you finish your last dose. Immunotherapy, however, aims to create a permanent change. It’s all about teaching your immune system a new trick—how to spot and destroy cancer cells for the long haul.
The Biggest Plus: Lasting Immune Memory
The single most powerful advantage of immunotherapy is its potential to create durable, long-term responses. Think of it as giving your immune cells a permanent education. The treatment can build something called “immune memory,” which means your body remembers what the cancer looks like, staying on guard to attack it if it ever dares to show up again, even years down the road.
For some people with very advanced cancers, this has led to remissions that last for years. It’s completely flipped the script for diseases that were once seen as a death sentence. A patient diagnosed with metastatic melanoma today, for instance, has a dramatically better chance at long-term survival than they did just over a decade ago, and that’s largely thanks to immunotherapy.
“Immunotherapy has fundamentally changed cancer medicine with the possibility of a cure for cancers that have been considered completely incurable… Today, a person with this diagnosis has a one in three chance of being cured. That’s a dramatic improvement in outcomes.”
This shot at lasting control is what makes immunotherapy such a game-changer in cancer care.
The Other Side of the Coin: Immune-Related Side Effects
But taking the brakes off your immune system isn’t always a perfectly precise operation. The goal is for your supercharged immune cells to go after only the cancer. Sometimes, though, they get a bit overzealous and start attacking healthy, normal tissues by mistake. When that happens, we get what are called immune-related adverse events, or irAEs.
These are a whole different beast from chemo side effects. They aren’t caused by a poison that kills cells. Instead, they’re the result of an overactive immune response—basically, inflammation firing up in parts of the body where it shouldn’t be.
The side effects can be all over the map, depending on which part of the body the immune system decides to target. Some of the most common reactions include:
- Skin Issues: Rashes, itchiness, and dry skin are very common. The skin is just one of those frequent targets for an activated immune system.
- Fatigue: This isn’t just feeling a little tired. It can be a deep, persistent exhaustion as your body pours a massive amount of energy into its immune fight.
- Gut Problems: Inflammation in the digestive tract can cause diarrhea or a more serious condition called colitis.
- Hormone Gland Issues: Sometimes the immune system targets glands that produce hormones, like the thyroid or pituitary, which can disrupt their normal function.
While that list can sound a bit scary, it’s helpful to remember that these reactions are often a sign that the treatment is doing its job and revving up your immune system.
Why Managing Side Effects Is Key to Success
The great news is that oncology teams are now incredibly skilled at spotting and managing these immune-related side effects. The absolute key is fast and open communication. Your doctors and nurses need to know about any new or worsening symptom as soon as it happens, no matter how small it might seem.
The approach to managing these reactions is pretty straightforward:
- Catch It Early: Constant monitoring and you reporting any changes are the first line of defense.
- Manage the Symptoms: For milder issues, simple things like creams for a rash or medication for diarrhea can keep things under control.
- Calm the Immune System: If a reaction is more serious, doctors can prescribe steroids or other drugs to temporarily dial back the immune response.
This proactive management allows most people to stay on their treatment safely and effectively. It’s all about finding that sweet spot: keeping the immune system fired up enough to fight the cancer, while making sure its effects on the rest of your body are well-managed.
Navigating the whole experience—both the physical and emotional sides—is a journey. Taking care of your overall well-being is a huge part of it. If you’re looking for strategies to support yourself, our guide on mental health and self-care tips helpful might offer some useful ideas.
Who Is a Good Candidate for Immunotherapy
Figuring out if immunotherapy is the right call isn’t a simple yes-or-no question. It’s a detailed conversation you’ll have with your oncology team, a decision that hinges on a unique mix of factors about you, your cancer, and even its specific genetic fingerprint.
Think of it as your oncologist playing detective. They need to gather all the clues to see if unleashing your immune system is the best strategy. It’s not a one-size-fits-all treatment, and the goal is always to tip the scales in favor of a strong response while keeping a close eye on any potential risks.
The Role of Cancer Type and Stage
The first and most important clue is the cancer itself. Immunotherapy has been a game-changer for certain cancers, but it doesn’t work the same way for every type. A lot of its success comes down to how “visible” a particular cancer is to your immune system.
Cancers with a high number of genetic mutations—like many melanomas or certain lung cancers—are often fantastic candidates. All those mutations create a bunch of abnormal proteins on the cancer cells, basically waving red flags that help the immune system recognize them as a threat. This gives immunotherapy a much clearer target.
The cancer’s stage also matters a great deal. Immunotherapy is often a go-to for advanced or metastatic cancers that have already spread. But we’re also seeing it used more and more in earlier stages, sometimes after surgery, to help hunt down any remaining cancer cells and stop them from coming back.
Biomarker Testing: The Compatibility Score
This is where the science gets really personal. Biomarker testing is probably the most critical piece of the puzzle. Imagine it as a “compatibility score” between your specific tumor and an immunotherapy drug. By analyzing a sample of your tumor tissue, doctors can look for molecular signs that predict how well the treatment might work.
One of the most important biomarkers they look for is a protein called PD-L1.
- What it is: You can think of PD-L1 as a “secret handshake” that cancer cells use to tell T-cells, “I’m one of you, move along.” It’s a trick to hide in plain sight.
- What testing measures: The test measures just how much of this protein is on your cancer cells. If there’s a lot of it, it’s a good sign the cancer is relying heavily on this invisibility cloak to survive.
- Why it matters: A high PD-L1 level is a strong hint that a checkpoint inhibitor drug, which is designed to block that very handshake, will likely be effective.
Other biomarkers, like tumor mutational burden (TMB) and microsatellite instability (MSI), also offer valuable insights. They help paint a picture of whether a tumor is the kind that’s likely to provoke a powerful immune response. This data-driven approach is what modern cancer care is all about.
“We’re working with what the body naturally does but has not done perfectly, as it has allowed cancer to develop. We’re trying to reinforce the body’s natural defenses.”
It’s a shift from treating “lung cancer” to treating your lung cancer, based on its unique biological playbook.
Considering Your Overall Health
Finally, your own health profile is a huge part of the conversation. Since immunotherapy is designed to supercharge your immune system, your general health and any pre-existing conditions need to be carefully considered.
Your oncologist will take a close look at your medical history, especially if you have any autoimmune diseases like rheumatoid arthritis, lupus, or Crohn’s disease. Because immunotherapy ramps up the entire immune system, there’s a chance it could make these conditions flare up.
Your overall physical fitness and how well your organs are working are also assessed. The team needs to be confident you can handle the treatment and manage any side effects that pop up. In the end, it’s always a shared decision, weighing the incredible potential of immunotherapy against the risks based on your specific situation.
Your Questions About Immunotherapy, Answered
A cancer diagnosis opens the door to a flood of questions. When immunotherapy enters the picture, it can feel like learning a whole new language. How does it work? What should I expect? What does this mean for me?
We’ve heard these questions from countless patients and their families. Our goal here is to cut through the jargon and give you clear, straightforward answers. Think of this as a starting point to help you feel more informed and ready for the road ahead.
How Is Immunotherapy Different From Chemotherapy?
The biggest difference between immunotherapy and chemotherapy comes down to their fundamental battle plans. They couldn’t be more opposite in how they take on cancer, which is why their side effects and the patient experience are so different.
Think of chemotherapy as a direct assault. It uses powerful drugs that are designed to hunt down and kill any cell in the body that’s dividing quickly. This works well against fast-growing cancer cells, but it’s not a targeted attack. It often causes collateral damage to healthy, fast-dividing cells, like the ones in your hair, the lining of your stomach, or your bone marrow.
Immunotherapy, on the other hand, is an indirect strategy. It doesn’t attack the cancer cells itself. Instead, it’s more like a training program for your own immune system, teaching it to recognize and wipe out cancer cells on its own. It’s like giving your body’s internal security team a new set of tools and better intel so they can finally do the job they were meant to do.
This is a critical distinction. The side effects from immunotherapy usually stem from an over-caffeinated immune response, not the kind of direct cellular damage we see with chemo.
How Long Does It Take for Immunotherapy to Work?
Patience is a big part of the immunotherapy journey. Unlike chemotherapy, where we might see results fairly quickly, immunotherapy works on a much more gradual timeline. That’s because it takes time to wake up your immune system and get it ready for battle.
First, the treatment needs to take the “brakes” off your T-cells. Then, those newly activated T-cells have to travel through your body, hunt down the tumor, identify it as a threat, and launch their attack. This whole process can take anywhere from several weeks to a few months before we can see a clear effect on a scan.
Your oncology team will be watching your progress carefully. It’s also good to know that sometimes, a tumor might look like it’s getting a little bigger on the first scan. This isn’t always bad news. This effect, called pseudoprogression, can actually be a great sign that a swarm of immune cells has rushed to the tumor and is actively attacking it, causing inflammation.
Can Immunotherapy Be Combined with Other Cancer Treatments?
Absolutely. In fact, combination therapy is quickly becoming the standard of care in modern oncology. Hitting the cancer from multiple angles at once often leads to far better results than relying on just one treatment. It’s like a coordinated military assault instead of a single-front attack.
Here are a few common tag-teams:
- With Chemotherapy: This can be a very powerful duo. Chemo can kill off a chunk of cancer cells, and as those cells die, they release proteins called antigens—think of them as “wanted posters.” This makes it much easier for the newly fired-up immune system to find and finish off any cancer cells left behind.
- With Radiation Therapy: Radiation damages a tumor’s DNA, causing it to send out distress signals that get the immune system’s attention. Pairing it with immunotherapy can help turn a local treatment (radiation) into a system-wide, body-wide response.
- With Targeted Therapy: These drugs go after specific molecules that cancer needs to grow. Using them alongside immunotherapy can weaken the cancer’s defenses, leaving it more exposed and vulnerable to an immune attack.
Is Immunotherapy a Cure for Cancer?
In the world of oncology, we use the word “cure” very carefully, but immunotherapy has brought us closer to that reality for more people than ever before. For some patients with advanced cancers that were once considered a death sentence, like melanoma or lung cancer, these treatments have produced what we call durable, long-term remissions.
What does that mean? It means the cancer goes away and stays away for years, even after the treatment stops. In these amazing cases, the patient’s own immune system has developed a “memory” of the cancer and continues to patrol the body, on the lookout for any signs of its return. Some doctors call this a “functional cure.”
However, it’s so important to understand that immunotherapy doesn’t work for everyone. Success rates can vary a lot depending on the type of cancer, its unique genetic markers, and the individual patient. For many, the goal is to turn cancer into a manageable, chronic condition or to achieve a lasting remission—giving people precious time and hope that was once unimaginable.
At maxijournal.com, we are committed to providing clear, accessible information on the topics that matter most to you, from health and science to technology and the arts. Explore our daily writing to stay informed and inspired.
Learn more and read our latest articles at maxijournal.com.
Discover more from Maxi Journal
Subscribe to get the latest posts sent to your email.