Stem cell therapy is a fascinating field that taps into your body’s innate ability to heal itself. At its heart, the treatment uses special cells—called stem cells—that act like a master repair kit, helping to regenerate and fix everything from damaged cartilage and bone to muscle and nerves.
Unpacking the Basics of Stem Cell Therapy
Think of your body as having a team of microscopic construction workers always on call. These are your stem cells, and they have two incredible superpowers: they can make endless copies of themselves, and under the right circumstances, they can transform into other types of specialized cells.
So, what does this mean in practice? If you have a torn muscle, a stem cell can become a new muscle cell. If the cartilage in your knee is worn down, a stem cell can become a new cartilage cell. This remarkable transformation is called differentiation, and it’s the entire basis for how stem cell therapy works.
To give you a quick overview, here’s a simple breakdown of the core ideas.
Stem Cell Therapy at a Glance
| Concept | Simple Explanation |
|---|---|
| Stem Cells | “Blank” or “master” cells that can turn into specialized cells (like skin, muscle, or bone). |
| Differentiation | The process where a stem cell transforms into a specific cell type to perform a certain job. |
| Regeneration | The body’s natural ability to repair or replace damaged tissue, which stem cells help drive. |
| Mechanism | Involves reducing inflammation, signaling other cells to help, and replacing damaged cells. |
This table shows how the therapy isn’t about introducing a foreign drug but rather about boosting a natural biological process.
The Master Repair Kit Analogy
A great way to picture this is to think of stem cells as a blank slate or a universal tool in a repair kit. They don’t start with a specific job, which gives them the amazing flexibility to adapt to whatever the body needs fixed.
When an injury or disease occurs, these cells are called to the site of the damage. Once there, they get to work in a few key ways:
- They reduce inflammation. Stem cells are fantastic at calming down the body’s inflammatory response, which is often a major source of pain and can get in the way of proper healing.
- They signal other cells to help. Think of them as a foreman on a job site. They release powerful growth factors and proteins that shout instructions to other local cells, telling them to get involved in the repair process.
- They promote regeneration. In some situations, they can directly replace damaged or dying cells, essentially building new, healthy tissue from the ground up.
This entire approach marks a shift away from just managing symptoms with medications. Instead, it aims to fix the root cause of the problem by helping the body quite literally rebuild itself.
At its core, stem cell therapy empowers the body’s own healing potential. It’s not about introducing a foreign substance but about amplifying a natural process that has been part of our biology all along.
A Rapidly Growing Field of Medicine
The potential here hasn’t gone unnoticed. Stem cell therapy has grown from a niche area of research into a significant medical sector. The global market, valued at USD 297 million in 2023, is projected to hit USD 18.13 billion by 2026. This incredible expansion is being driven by new cell engineering technologies and growing enthusiasm for regenerative medicine, as detailed in recent market reports about stem cell therapy.
Getting a handle on these fundamentals is the first step. By learning how to guide these cellular powerhouses, doctors are opening new doors for treating conditions that were once considered chronic or untreatable.
What Kinds of Stem Cells Are There?
It turns out, not all stem cells are created equal. In regenerative medicine, we work with a few key types, and each one has its own unique origins, strengths, and jobs it’s best suited for. Getting a handle on these differences is the first step to understanding what stem cell therapy can actually do.
Think of it like a repair crew for your body. Some cells are master craftsmen who can build anything from scratch, while others are more like skilled specialists trained for a specific set of tasks. The three main categories you’ll hear about are embryonic stem cells, adult stem cells, and a fascinating lab-created type called induced pluripotent stem cells.
Each one has a distinct role to play, though for a mix of practical and ethical reasons, most of the treatments you’ll see today rely on adult stem cells.
Embryonic Stem Cells: The Ultimate Blank Slates
Embryonic stem cells (ESCs) are the most versatile of them all. They come from the inner cell mass of a blastocyst, which is a very early-stage embryo only a few days old. These cells are pluripotent, a term that means they hold the incredible potential to develop into any of the 200-plus cell types that make up a human being.
Imagine a sculptor having a single block of clay that could be molded into a heart, a nerve, or a skin cell—that’s the power of an embryonic stem cell. Their ability to form literally any tissue makes them a goldmine for research, letting scientists watch human development and disease unfold in ways that were once impossible.
But you’ll almost never see them used in a clinic. Their use is incredibly limited and fraught with ethical debate over their source. This controversy has pushed researchers to find alternatives that don’t involve human embryos.
Adult Stem Cells: The Body’s Own Repair Crew
You have stem cells in you right now. Found in various tissues and organs, adult stem cells are the body’s built-in repair system, constantly working to replenish and heal. Unlike their embryonic cousins, they are multipotent, which means they can turn into a limited range of cell types, usually those related to the tissue where they live.
Here are a couple of key examples:
- Mesenchymal Stem Cells (MSCs): These are found in bone marrow, fat (adipose) tissue, and umbilical cord tissue. They have the instructions to become bone, cartilage, muscle, and fat cells.
- Hematopoietic Stem Cells (HSCs): Also hanging out in bone marrow, these are the factory for our blood and immune systems. They are responsible for making all our red blood cells, white blood cells, and platelets.
A huge advantage here is that adult stem cells can be taken directly from your own body (this is called an autologous source). When your own cells are used, the risk of your immune system rejecting them drops to zero. This incredible safety profile is why MSCs from bone marrow and fat are the go-to choice for many of today’s regenerative therapies, especially for things like arthritis and sports injuries.
Using a patient’s own adult stem cells is the cornerstone of modern regenerative medicine. We’re essentially taking the body’s natural repair kit, concentrating it, and putting it right where it’s needed most.
Induced Pluripotent Stem Cells: The Scientific Breakthrough
What if you could take a regular cell, like one from your skin, and rewind its biological clock until it acted just like an embryonic stem cell? It sounds like science fiction, but that’s exactly what induced pluripotent stem cells (iPSCs) are. This discovery was so profound it won a Nobel Prize in 2012, and it has completely changed the game.
Scientists use specific proteins to “reprogram” a specialized adult cell, forcing it to revert to a pluripotent state. Once reprogrammed, it can once again become any cell type in the body, just like an ESC.
The benefits are massive:
- No Ethical Issues: Because they start as adult cells (like skin or blood), they sidestep the entire ethical debate surrounding embryos.
- Perfectly Personalized: iPSCs are made from a patient’s own cells, creating a perfect genetic match for transplantation and eliminating any fear of immune rejection.
- A Window into Disease: Researchers can take cells from patients with genetic disorders, turn them into iPSCs, and then grow them into specific tissues in a petri dish. This lets them watch how a disease develops and test new drugs on real human tissue without any risk to the patient.
Right now, iPSCs are mostly found in research labs and early-stage clinical trials, but they represent a huge leap forward toward a future of truly personalized medicine.
How Stem Cell Therapy Actually Works in Your Body
Ever wonder what really happens after a stem cell injection? It’s less about a magic bullet and more about deploying a highly skilled biological repair crew right where your body needs it most. The whole process is surprisingly straightforward, but the science behind it is fascinating.
It all starts with a detailed consultation. Your doctor will review your medical history and imaging to create a precise treatment plan, much like an architect drafts a blueprint before construction begins. This ensures the therapy is tailored specifically to your injury.
Next comes the collection, or “harvesting,” of the stem cells. In most cases today, this involves a simple procedure done under local anesthesia to draw cells from your own bone marrow or adipose (fat) tissue. Using your own cells—what we call an autologous approach—virtually eliminates the risk of rejection. Your body recognizes them as its own.
Once the cells are collected, they’re not ready to be re-injected just yet. They head to the lab for preparation, where technicians isolate and concentrate the stem cells. Think of this as assembling the most elite members of the repair crew, making sure you have the highest concentration of active cells ready to get to work.
The process boils down to a few key stages:
- Step 1: Harvest: A small sample of your bone marrow or fat is collected to source your body’s own repair cells.
- Step 2: Preparation: The sample is processed to isolate and concentrate the powerful stem cells, maximizing their potential.
- Step 3: Injection: The concentrated stem cells are carefully injected back into your body directly at the site of injury, often using ultrasound for pinpoint accuracy.
Once inside your body, the stem cells act like project managers on a construction site. They don’t just turn into new tissue themselves; they orchestrate the entire healing process. They do this largely through something called the paracrine effect—releasing a cascade of signals that tell other cells in the area what to do.
First, they work to calm inflammation, which is a bit like the crew clearing away debris and rubble from a damaged structure before they can start rebuilding. This initial action is often what leads to a rapid reduction in pain and sets the stage for meaningful, long-term tissue regeneration.
Visualizing the Different Types of Stem Cells
Not all stem cells are the same. The graphic below shows the three main types you’ll hear about, each with unique capabilities.
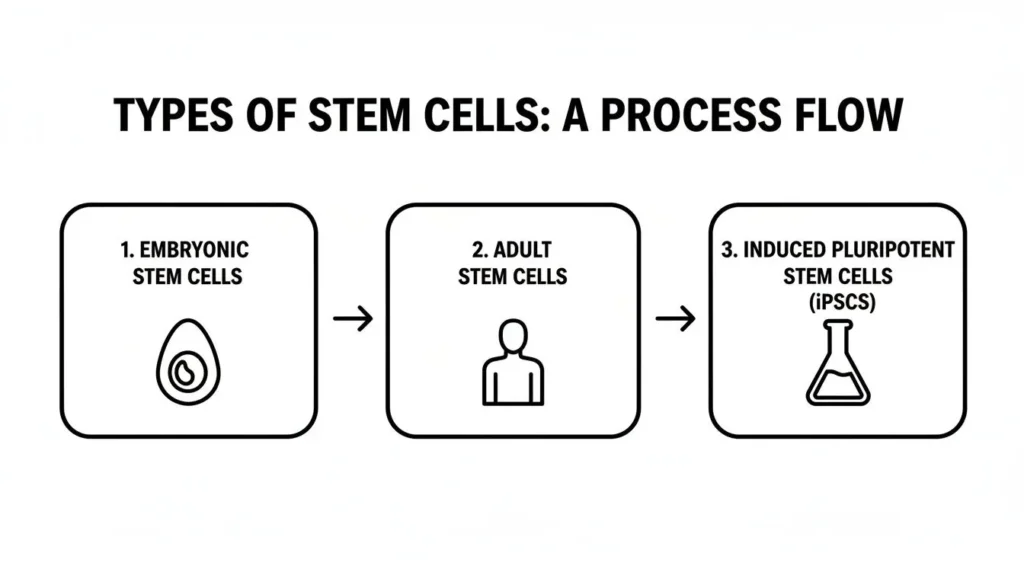
From the “blank slate” potential of embryonic cells to the specialized adult cells we have in our bodies and the remarkable lab-created iPSCs, each type offers different strengths for different therapeutic needs.
The field has seen explosive growth. The number of providers jumped from just 772 in 2017 to an estimated 1,550 by 2025. Today, adult stem cells are the workhorses of the industry, projected to account for 86.1% of the revenue share by 2026. If you want to dive deeper, you can read the full research about the growth of regenerative medicine services on PMC NCBI.
After being deployed, the stem cells get to work in three primary ways:
- They direct immune cells to manage the local environment, clearing out damaged tissue and reducing harmful inflammation.
- They release a cocktail of growth factors that stimulate the formation of new blood vessels and encourage nearby cells to start rebuilding.
- In some cases, they can differentiate—or transform—into the specific cell types needed to replace what was lost, like new cartilage or bone cells.
This is where the repair crew analogy really comes to life. Your stem cells are the managers, signaling to other local cells and guiding them to rebuild the tissue from the ground up, ensuring it’s done correctly.
Coordinating the Entire Healing Response
Beyond just building new tissue, stem cells are master coordinators. They improve blood flow to the injured area and continue to modulate inflammation, creating the perfect environment for healing.
This means swelling subsides faster, and a steady supply of oxygen-rich blood and nutrients can reach the site to fuel the repair process.
Key Insight: Stem cells don’t just replace damaged cells; they are biological conductors, orchestrating a complex symphony of signals that guide the entire repair process from start to finish.
With these signals in place, new blood vessels form, nutrients flood the area, and structural proteins begin to assemble—like scaffolding going up around a building, providing the framework for a solid reconstruction. As the area matures, the stem cells may then differentiate into the final cell type needed, depending on the cues from their surroundings.
To ensure the best possible outcomes, reputable clinics adhere to strict protocols:
- Proper Handling: Cells are handled with extreme care to preserve their viability and potency.
- Guided Delivery: Imaging technology like ultrasound is used to guarantee the cells are placed exactly where they’re needed most.
- Post-Procedure Monitoring: Your progress is tracked carefully to ensure you are healing as expected.
Ultimately, stem cell therapy is about empowering your body’s innate ability to heal itself. It’s not an external fix; it’s a way of concentrating your own natural resources and putting them right where they can do the most good. If you’re exploring this option, remember that these cells are your on-site managers, demolition crew, and builders all rolled into one.
Real-World Applications and Promising Research
The true power of stem cells comes into focus when we look at how they’re being used today—and where the research is headed tomorrow. It’s a field with two distinct sides: on one hand, we have established, FDA-approved treatments that are a standard part of medical care. On the other, we have a whole world of promising experimental therapies that are pushing the boundaries of what we thought was possible.
Knowing the difference is critical. Some applications have decades of proven success behind them, while others are still in the early stages, offering hope but not yet a guaranteed cure.
Established and Approved Stem Cell Therapies
The most famous and widely accepted use of stem cells is hematopoietic stem cell transplantation (HSCT), which most people know as a bone marrow transplant. For over 50 years, this procedure has been a literal lifesaver for people with certain blood cancers, like leukemia and lymphoma, and blood disorders such as sickle cell anemia.
The process is intense but elegant. A patient’s diseased bone marrow is first cleared out using chemotherapy or radiation. Then, they receive an infusion of healthy hematopoietic stem cells—either from a compatible donor or from their own cells collected earlier. These new cells find their way to the bone marrow and get to work, producing a fresh supply of healthy blood cells. It’s a powerful, real-world example of using stem cells to completely rebuild a vital system in the body.
These treatments are considered the gold standard for a few key reasons:
- Proven Efficacy: Decades of clinical data confirm they work for specific conditions.
- Standardized Protocols: The methods for harvesting, preparing, and transplanting these cells are highly regulated and consistent.
- FDA Approval: They have passed rigorous testing and earned official approval from agencies like the U.S. Food and Drug Administration.
Promising Areas of Experimental Research
So, what about everything else? Beyond bone marrow transplants, researchers are exploring how different kinds of stem cells, particularly Mesenchymal Stem Cells (MSCs), could treat a massive range of other conditions. These are the applications you often hear about in the news—they’re currently considered experimental, meaning they’re being studied in clinical trials but aren’t yet approved as standard treatments.
One of the most active areas of research is orthopedics. You’ve probably seen clinics offering stem cell injections for things like osteoarthritis, tendonitis, and ligament tears. The idea here is to use the cells’ powerful anti-inflammatory and regenerative signaling abilities to reduce pain, improve joint function, and maybe even delay the need for a full-blown joint replacement surgery.
But the research doesn’t stop there. Scientists are looking at stem cells for some of the most complex diseases we face:
- Neurological Conditions: For diseases like Parkinson’s, multiple sclerosis (MS), and spinal cord injuries, the hope is that stem cells can protect the nerve cells we have and perhaps even replace those that have been lost.
- Cardiovascular Disease: After a heart attack damages the heart muscle, stem cells might be used to help repair that tissue and improve the heart’s ability to pump.
- Autoimmune Diseases: In conditions like Crohn’s disease or rheumatoid arthritis, stem cells may help “reset” the immune system, calming its attack on the body’s own tissues.
To give you a clearer picture, here’s a quick breakdown of where things stand for various conditions.
Status of Stem Cell Therapy for Various Conditions
This table shows the difference between established, FDA-approved treatments and promising but still experimental applications. It’s a great way to understand the current landscape.
| Condition | Treatment Status | Description of Application |
|---|---|---|
| Leukemia & Lymphoma | Established & FDA-Approved | Hematopoietic Stem Cell Transplants (HSCT) are a standard of care to restore bone marrow after chemotherapy/radiation. |
| Sickle Cell Anemia | Established & FDA-Approved | HSCT can provide a potential cure by replacing faulty blood-producing cells with healthy ones from a donor. |
| Osteoarthritis | Experimental | Mesenchymal Stem Cell (MSC) injections are being studied to reduce inflammation, manage pain, and potentially regenerate cartilage. |
| Multiple Sclerosis (MS) | Experimental | Researchers are investigating if HSCT can “reboot” the immune system and if MSCs can reduce inflammation and promote nerve repair. |
| Parkinson’s Disease | Experimental | Clinical trials are exploring whether stem cells can be differentiated into dopamine-producing neurons to replace those lost to the disease. |
| Heart Disease | Experimental | Studies are focused on using stem cells to repair heart muscle damaged by a heart attack and improve overall cardiac function. |
| Spinal Cord Injury | Experimental | The goal is to use stem cells to reduce inflammation, protect surviving nerve cells, and encourage new neural connections. |
While many of these applications are still under investigation, the breadth of research shows just how much potential scientists see in this field.
The versatility of stem cells has made them a focal point of modern medicine. Research is expanding to address acute illnesses, not just chronic ones, showcasing their potential to adapt to urgent medical needs.
We saw this adaptability in action during the COVID-19 pandemic. A 2021 study involved giving stem cell infusions to patients with severe lung damage from the virus. The results were promising, with no negative side effects reported, showing how this therapy could be applied in critical care situations. If you want to dive deeper into the market trends, you can discover more insights about the expanding stem cell therapy market on novaoneadvisor.com.
The Future of Regenerative Healing
Looking ahead, the field is moving toward even more sophisticated uses. Scientists are exploring ways to combine stem cell therapy with other technologies, like gene editing. This could one day allow us to correct genetic defects in a patient’s own cells before putting them back into the body. If you find that fascinating, you might want to check out our guide on how CRISPR works.
Ultimately, the goal is to shift from just managing symptoms to truly restoring function. While a lot of this is still in the research phase, the progress we’re seeing is paving the way for a new era of medicine—one where we can help the body heal itself from the inside out.
Sorting Through the Promise, Perils, and Pitfalls

The buzz around stem cell therapy is powerful, and for good reason. The potential benefits—from real pain relief to getting back on the tennis court—are compelling, especially for people dealing with stubborn orthopedic problems.
But getting the full story means looking past the hype. Like any medical procedure, stem cell therapy isn’t a magic bullet, and it comes with its own set of risks.
Even when everything is done by the book, there are potential downsides. You could face an infection at the injection site, deal with some temporary pain and swelling, or, if you’re receiving cells from a donor (allogeneic therapy), there’s a chance your body’s immune system could react.
Knowing this is step one. Step two is understanding who’s watching the watchers: the regulatory bodies like the U.S. Food and Drug Administration (FDA).
Regulation: Your Best Defense Against Bad Actors
The FDA’s entire mission is to confirm that medical treatments actually work and won’t harm you. As it stands, the only stem cell products with the FDA’s full seal of approval are those made from hematopoietic (blood-forming) stem cells, which are primarily used to treat certain blood cancers and disorders.
Most other stem cell therapies, especially those you hear about for joints or autoimmune diseases, are still in the investigational stage. Clinics can offer them, but they have to play by a strict set of rules. A big one? They can’t legally claim their treatment will cure, fix, or even prevent a specific disease.
Here’s the bottom line: Regulation exists to protect you from empty promises and dangerous procedures. If a clinic guarantees a cure or says their therapy is a sure thing for your condition, they’re stepping way outside of established medical and ethical lines.
Unfortunately, this regulatory gray zone has paved the way for predatory clinics and “stem cell tourism,” where people travel for unproven treatments that can be both ineffective and unsafe.
How to Tell a Good Clinic from a Bad One
Learning to spot a trustworthy medical provider versus a high-pressure sales pitch is probably the most important skill you can have. A reputable clinic always puts your safety first and is completely transparent. They’ll start with a deep dive into your medical history and a thorough evaluation, including imaging, to see if you’re even a good candidate. For a clearer understanding of how medical fields evolve, you might be interested in our article explaining what immunotherapy is for cancer.
On the flip side, certain red flags should make you walk away immediately. Be suspicious of any clinic that bombards you with aggressive ads, uses patient testimonials as their main proof, or pushes you to sign up on the spot. Real medicine doesn’t come with a “limited-time offer.”
Here are the critical questions you should ask any clinic you’re considering:
- What kind of cells are you using? They should tell you the exact source, like your own bone marrow or adipose (fat) tissue.
- How do you process the cells? Look for adherence to strict sterile procedures, often called cGMP (Current Good Manufacturing Practice) standards.
- What are the real risks of this procedure? An honest doctor will have a frank conversation with you about every possible complication.
- Can you show me the scientific evidence for my specific condition? They should be able to point to actual clinical studies, not just a handful of success stories.
Armed with the right questions and a healthy dose of skepticism, you can cut through the noise and make a choice that’s truly best for your health, grounded in real science and realistic outcomes.
What the Future Holds for Regenerative Healing
Looking at the horizon, it’s clear stem cell therapy isn’t just a niche field anymore; it’s on track to become a fundamental part of mainstream medicine. The research happening right now is building the foundation for a future where we don’t just put a bandage on chronic diseases, but actually repair the damage they leave behind.
We’re moving past simple injections and into a realm of highly sophisticated, targeted treatments that could completely change our understanding of what it means to heal.
This evolution is fueled by combining stem cells with other powerful technologies. For example, scientists are exploring how to merge stem cell therapy with gene-editing tools. Imagine taking a patient’s own cells, correcting the genetic flaw causing their illness, and then reintroducing those healthy, repaired cells back into their body. If that sounds fascinating, you can dive deeper into our guide on what gene therapy is and how it works.
The Next Wave of Innovation
What’s coming in the next decade might sound like science fiction, but it’s grounded in real, ongoing research. The focus is squarely on creating treatments that are more effective, more accessible, and truly personalized.
Here are a few key areas to keep an eye on:
- 3D Bioprinting: Think of stem cells as a kind of biological ink. Scientists are already using them to print three-dimensional tissues and are working their way toward simple organs. This could one day mean custom-made replacement parts for the body, making donor waiting lists a thing of the past.
- “Off-the-Shelf” Therapies: One of the biggest goals in the field is to develop universal stem cell lines. These cells could be grown in huge batches, stored, and be ready to use for any patient at a moment’s notice. This would slash costs and make treatments available on demand.
- Smarter Delivery Systems: The future isn’t just about the cells, but how we get them where they need to go. Researchers are developing advanced methods—like using special biomaterials or hydrogels—that act as a scaffold, guiding the stem cells and helping them integrate into the surrounding tissue.
The ultimate vision for regenerative medicine is to transition from merely treating symptoms to restoring full biological function. This involves not just replacing what’s lost but recreating a healthy, self-sustaining environment within the body.
Of course, this future isn’t here just yet. Rigorous clinical trials are still the bedrock of this field, ensuring every new advancement is both safe and genuinely effective. But the forward momentum is undeniable.
The very concept of stem cell therapy is shifting from a highly specialized procedure to a foundational tool for a new era of medicine. It holds the potential for a future where we can truly regenerate, repair, and restore the body from the inside out.
Your Top Questions About Stem Cell Therapy Answered
When you’re considering a treatment like stem cell therapy, the practical questions are often the first ones that come to mind. What will it feel like? How soon will I see a change? Let’s break down some of the most common questions to give you a real sense of the patient experience.
Is Stem Cell Therapy Painful?
Most people are pleasantly surprised by how manageable the process is. The level of discomfort really depends on where the cells are coming from.
If cells are taken from your own bone marrow or fat, that part is done under local anesthesia. You might feel a bit of deep pressure during the procedure and some soreness, like a bruise, for a few days after. As for the injection itself, it’s usually no more uncomfortable than a standard cortisone shot.
The Bottom Line on Comfort: The entire process is designed to be as comfortable as possible. Think of it as a minimally invasive procedure, not major surgery. Any discomfort is typically mild and short-lived.
Your medical team’s top priority is your comfort, and they’ll guide you through every step of the way.
How Long Does It Take to See Results?
This is the big one, and it’s important to set the right expectations. Stem cell therapy isn’t a quick fix; it’s about kick-starting your body’s own repair mechanisms, and that process takes time.
While some people notice a drop in pain and inflammation within the first few weeks, the real magic happens over a longer period. Meaningful tissue repair and functional improvements usually become noticeable between three to six months after the treatment. Think of it as planting a seed—it needs time to grow and strengthen from the inside out.
What’s the Difference Between Autologous and Allogeneic Cells?
This question is all about where the stem cells originate. It’s a simple distinction but a crucial one for your treatment plan.
- Autologous Therapy: This means the cells come from you. They’re typically collected from your own bone marrow or fat tissue. The huge advantage here is that there’s zero risk of immune rejection. Your body knows these cells are its own.
- Allogeneic Therapy: This means the cells come from a healthy, thoroughly screened donor. Umbilical cord tissue is a common source for these. While donor cells can be incredibly powerful, there’s a small risk of an immune response, which your doctor will carefully monitor and manage.
The best approach for you will depend on your health, your age, and the specific condition being treated.
Is Stem Cell Therapy Covered by Insurance?
For the most part, insurance coverage is still playing catch-up with the science. Insurers generally stick to covering procedures that have full FDA approval, like bone marrow transplants for treating certain cancers.
Unfortunately, many of the more common applications—especially for orthopedic conditions like arthritis or sports injuries—are often classified as “investigational.” This usually means the cost falls to the patient. It is absolutely essential to have a frank conversation about the full cost with any clinic you’re considering and to call your insurance provider directly. Getting clarity on the financial side upfront is key to avoiding any surprises down the road.
At maxijournal.com, we publish clear, well-researched articles on science, health, and technology to keep you informed. Explore more topics at https://maxijournal.com.
Discover more from Maxi Journal
Subscribe to get the latest posts sent to your email.