Nanotechnology is opening up a whole new frontier in medicine, giving us microscopic tools to diagnose and treat diseases with a level of precision we’ve only dreamed of. This field, which we often call nanomedicine, works at the scale of individual cells and molecules—a scale once reserved for science fiction. It’s all about engineering incredibly tiny particles to deliver drugs right where they’re needed, spot illnesses earlier than ever, and even help our bodies regenerate damaged tissue.
How Nanotechnology Is Remaking Modern Medicine
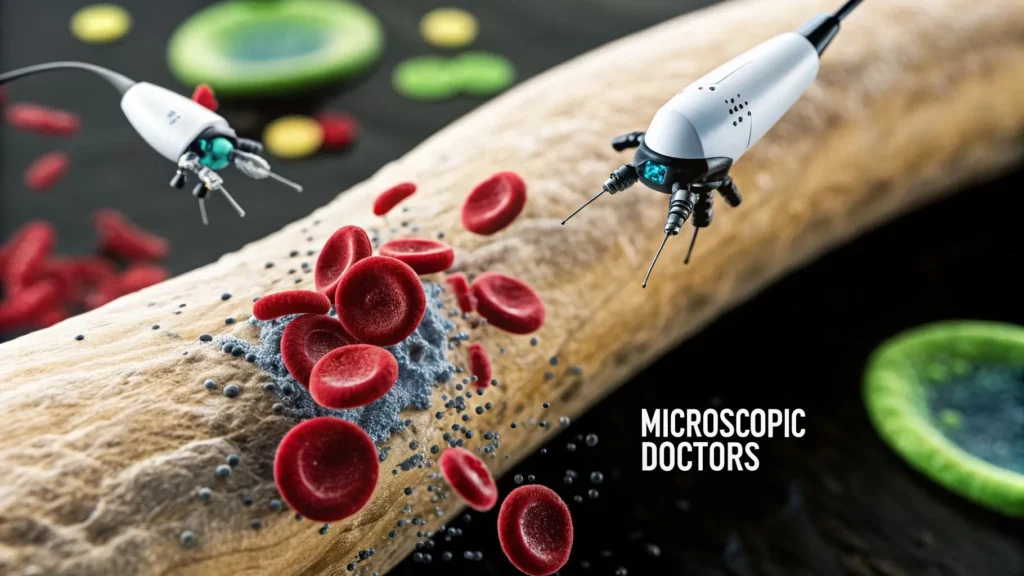
Picture a team of microscopic doctors, so small they can navigate your bloodstream to find and repair damaged cells one by one. That’s the essential promise of nanomedicine. It involves designing and using materials at the nanoscale—typically between 1 and 100 nanometers—to interact with our biology in powerful new ways.
To give you a sense of just how small that is, a single human hair is about 80,000 nanometers wide. Working at this level allows scientists to build tools that are actually smaller than our own cells.
The Power of Being Small
The real game-changer with nanotechnology is its incredibly small size. Particles engineered at this scale can get into parts of the body that conventional drugs and larger molecules just can’t reach. They can sneak across the blood-brain barrier to treat neurological diseases or slip inside a cancer cell to deliver a fatal dose of medication without harming the healthy tissue around it.
Think of nanoparticles as ‘smart keys’ designed to unlock specific medical treatments inside the body. Each key can be custom-built to fit only one type of lock—like a specific disease marker on a cell’s surface. This kind of precision targeting is what makes nanotechnology applications in medicine so powerful.
At its heart, nanomedicine is about doing more with less. It opens the door to more effective treatments with fewer side effects, completely changing the risk-benefit equation for patients facing serious illnesses like cancer, heart disease, and neurodegenerative disorders.
A Field on the Rise
We’re quickly moving from futuristic ideas to practical, real-world applications, and the market growth reflects that. In 2021, the global nanomedicine market was valued at $242.6 billion, and it’s projected to rocket to $493.5 billion by 2026. This boom is largely fueled by the success of nanoparticle-based drug delivery systems; in fact, over 50 such products are already approved for clinical use, with hundreds more in the pipeline. You can dig deeper into these market trends and what they mean for the future of healthcare.
This table gives you a quick snapshot of the core areas where nanomedicine is already making waves.
Core Nanotechnology Applications in Medicine
| Application Area | Main Objective | Core Technology Example |
|---|---|---|
| Drug Delivery | Deliver high concentrations of drugs directly to diseased cells, minimizing side effects. | Liposomes or polymer-based nanoparticles that encapsulate a drug payload. |
| Diagnostics | Detect disease biomarkers at extremely low concentrations for earlier diagnosis. | Quantum dots or gold nanoparticles that bind to specific molecules and emit a signal. |
| Imaging | Enhance the visibility of tissues or tumors in medical scans like MRI or CT. | Magnetic nanoparticles used as contrast agents to improve image clarity. |
| Tissue Engineering | Create scaffolds that mimic the body’s natural matrix to support cell growth and repair. | Nanofiber scaffolds that guide tissue regeneration for organs or skin. |
| Theranostics | Combine diagnostic and therapeutic functions into a single nanoparticle agent. | A nanoparticle that both highlights a tumor on a scan and delivers a drug to it. |
| Antimicrobial | Kill or inhibit the growth of bacteria and viruses on surfaces or in the body. | Silver or zinc oxide nanoparticles that disrupt microbial cell membranes. |
Each of these applications leverages the unique properties of nanomaterials to solve long-standing medical challenges in a new way.
Throughout this guide, we’ll walk you through a clear roadmap of the most important nanotechnology applications in medicine, exploring how they work and the real-world impact they’re having today. We’ll cover the key areas where this technology is making a difference, including:
- Targeted Drug Delivery: Delivering potent medicines directly to diseased cells.
- Early Disease Diagnostics: Spotting illnesses at their most treatable stages.
- Regenerative Medicine: Using nanomaterials to help the body heal itself.
- Theranostics: Combining diagnosis and therapy into a single, powerful tool.
Delivering Drugs with Pinpoint Accuracy
One of the biggest wins for nanotechnology in medicine has been in drug delivery. Think about traditional treatments like chemotherapy. They often work like a sledgehammer, flooding your entire system with powerful drugs. The goal is to kill diseased cells, but this approach causes a ton of collateral damage to healthy tissue, which is why the side effects can be so rough.
Nanotechnology offers a much smarter, more precise alternative.
Imagine a fleet of microscopic, programmable cargo ships, each loaded with a potent medicine. Instead of just dumping their contents everywhere, these ships are given a specific address: a tumor, an inflamed joint, or a pocket of infection. That’s the whole idea behind targeted drug delivery. We’re using nanoparticles as “medical smart bombs” designed to seek and destroy a specific target while leaving healthy cells alone.
This incredible precision comes from engineering the nanoparticle’s surface. Scientists can attach special molecules, like antibodies, that act like tiny homing beacons. These molecules are chosen because they only bind to specific receptors on the surface of diseased cells—kind of like a key that only fits one particular lock.
How Nanoparticles Act as Tiny Cargo Ships
Once these nanoparticles hit the bloodstream, they start to circulate. Thanks to their custom-designed surfaces, they pretty much ignore healthy cells. But when one bumps into its target—say, a cancer cell—the homing molecules lock on, and the nanoparticle gets to work.
This targeted drop-off allows for a much higher concentration of medicine right where it’s needed. It’s a game-changer. Some studies have shown that traditional chemotherapy struggles to get even 1% of the drug payload to the actual tumor. Nanoparticles can blow that ratio out of the water, making treatments far more effective at lower overall doses.
There are a few different types of nanoparticles that are common workhorses for this job, each with its own specialty:
- Liposomes: These are tiny, hollow spheres made from fat molecules, very similar to our own cell membranes. They’re great for carrying all sorts of drugs and are easy to customize for targeting.
- Polymer-based Nanoparticles: Built from biodegradable polymers, these particles are super versatile. You can design them to release their drug payload slowly over time, which is perfect for sustained treatment.
- Dendrimers: These have a unique, tree-like structure. All those “branches” give you tons of attachment points for drug molecules, making them incredibly efficient carriers.
- Inorganic Nanoparticles: Materials like gold or iron oxide are also in the mix. They’re especially useful because they can be designed to release their drug cargo when you hit them with an external trigger, like light or a magnetic field.
Real-World Impact in Cancer Treatment
This isn’t just theory; this technology is already in the clinic, making a real difference in cancer treatment. For years, approved nanoparticle-based chemotherapy drugs have been helping patients. They work by wrapping up toxic chemo agents in a protective shell, shielding the rest of the body from their harsh effects until they arrive at the tumor.
This doesn’t just cut down on side effects like hair loss and nausea; it also lets doctors use more powerful drug combinations that would otherwise be too toxic.
The ultimate goal of targeted drug delivery is to transform harsh systemic treatments into precise, localized therapies. It shifts the focus from broadly attacking a disease to surgically striking it at the molecular level, improving both patient outcomes and quality of life.
This kind of precision is also a key part of other advanced treatments. To get a better sense of how we’re teaching the body’s own defenses to join the fight, check out our guide on what is immunotherapy for cancer.
By turning nanoparticles into sophisticated delivery vehicles, we can make sure powerful medicines go only where they need to. It’s an approach that minimizes harm, maximizes effectiveness, and marks a huge leap forward in treating some of the world’s toughest diseases.
Diagnosing Diseases Earlier Than Ever Before
Beyond delivering treatments, one of the most exciting frontiers for nanotechnology in medicine is diagnostics. When it comes to diseases like cancer, Alzheimer’s, or heart conditions, catching them early is everything. Nanotechnology is essentially giving doctors a new kind of magnifying glass to spot these illnesses at their absolute earliest stages, often well before a person even feels sick.
You can think of nanoparticles as microscopic “biological detectives.” We can engineer these tiny agents to be incredibly sensitive biosensors, designed to patrol the bloodstream and pick up on the faintest chemical trails a disease leaves behind. They’re so precise they can identify specific biomarkers—like unique proteins or bits of genetic material linked to an illness—from just a single drop of blood.
This concept of engineering nanoparticles for specific jobs is central to nanomedicine, whether for targeting disease or delivering a drug.
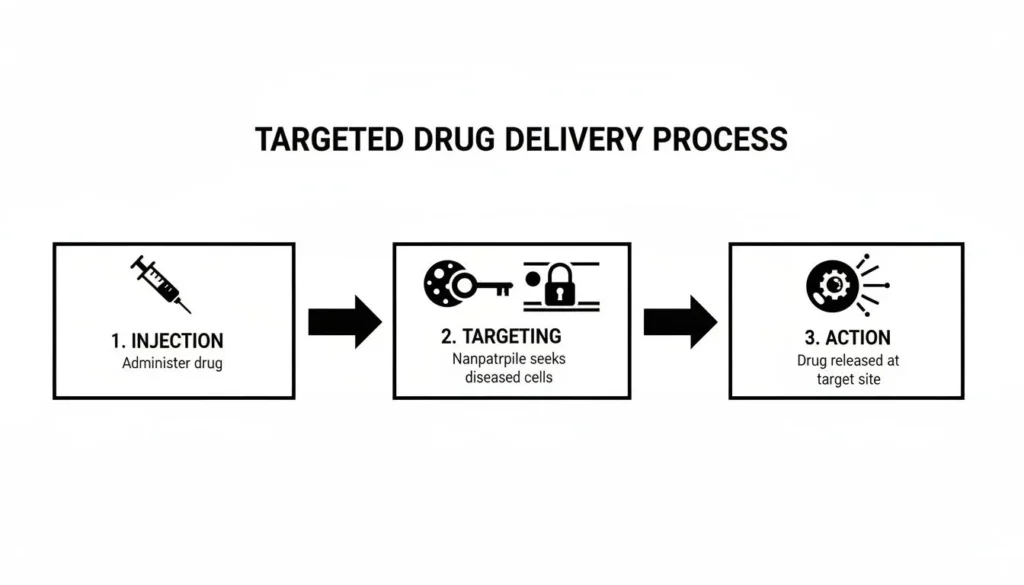
As you can see, the process is logical and direct: the nanoparticle is introduced into the system, it finds its specific biological target, and then it gets to work.
Enhancing Medical Imaging
Nanoparticles aren’t just finding biomarkers; they’re also making our medical imaging tools much more powerful. By acting as highly advanced contrast agents, they make scans like MRIs and CTs dramatically clearer and more detailed.
Imagine them as tiny light bulbs that doctors can strategically switch on inside the body. After being injected, these nanoparticles are designed to gather in specific areas of interest, like a tumor or inflamed tissue. Once there, they amplify the signal the scanner picks up, making diseased areas light up like a beacon on the screen. This allows radiologists to see abnormalities that would have been completely invisible with older methods.
This boost in visibility is a game-changer. It means doctors can find tumors when they’re much smaller and easier to treat, more accurately track how a disease is progressing, and see in real time if a treatment is actually working.
From the Lab to Clinical Practice
This isn’t just theory anymore. The technology is actively moving from the research bench into clinics, making diagnostics faster, cheaper, and more widely available. A fantastic real-world example is the use of gold nanoparticles in rapid diagnostic tests, which work on a similar principle to at-home pregnancy tests.
These tests use small strips coated with nanoparticles that are engineered to bind to a specific biomarker. When a sample like blood or saliva is added, the presence of that biomarker makes the nanoparticles cluster together. This clumping action creates a visible color change, giving a clear result in just a few minutes, no complex lab equipment needed.
- Speed: Get results in minutes, not days.
- Sensitivity: Detect disease markers even at very low concentrations.
- Cost-Effectiveness: Cut down the need for expensive, centralized laboratory machines.
- Accessibility: Make testing possible in remote areas or low-resource settings.
By creating diagnostic tools that are both more sensitive and more portable, nanotechnology is starting to level the playing field in healthcare. It’s arming doctors with better, faster information, which naturally leads to earlier interventions and better outcomes for patients. The massive amounts of complex data these new tools generate are often crunched using computational methods. If you’re curious about that side of things, you can learn more in our guide on what is bioinformatics. This powerful combination of nanotechnology and data science is truly pushing medical diagnostics into a new era.
Rebuilding The Body with Regenerative Medicine
Beyond just fighting disease, some of the most exciting work in medical nanotechnology involves helping the body heal and rebuild itself. This is the world of regenerative medicine, where scientists are essentially creating microscopic “construction crews” to repair damaged tissues from the inside out.

It’s a big shift away from just managing symptoms or swapping out damaged parts for artificial ones. The real goal here is to restore the body’s natural function by giving it the right tools and signals to heal properly.
Building Scaffolds for New Tissue
A core technique in this field involves creating nanofiber scaffolds. You can think of these as the perfect, tiny blueprint for growing new tissue. They’re built from biocompatible materials and woven into a structure that perfectly imitates the body’s own extracellular matrix—the natural support system our cells live in.
When one of these scaffolds is placed at an injury site, like a broken bone or torn cartilage, it gives the body’s own cells a structure to grab onto. From there, they can multiply and organize themselves into new, healthy tissue. The nanoscale design is what makes it work; it creates a surface that cells instinctively recognize and respond to, guiding the healing process far more effectively than a simple flat surface ever could.
To really kick the process into high gear, these scaffolds can be loaded up with growth factors or other signaling molecules.
These molecules are like foremen on a construction site, giving cells specific instructions like “grow here,” “form bone tissue,” or “create new blood vessels.” The nanoparticles in the scaffold are designed to release these instructions at just the right time and place, orchestrating a controlled and highly efficient healing process.
Making Medical Implants Safer and Smarter
Nanotechnology is also completely changing the game for medical implants like artificial joints, dental implants, and cardiovascular stents. One of the biggest hurdles with any foreign object in the body is the risk of infection or rejection.
Nanocoatings offer a pretty elegant solution. By applying an incredibly thin layer of antimicrobial nanoparticles—like silver or zinc oxide—to an implant’s surface, we create a powerful shield against bacteria. These nanoparticles kill microbes on contact, dramatically cutting down the risk of post-op infections, which are a huge reason implants fail. This kind of tech is foundational for modern bio-integrated devices. For those curious about how we manipulate biological building blocks on a genetic level, it’s worth learning about how CRISPR works and its own role in regenerative science.
On top of that, nanotechnology helps implants become one with the body.
- Improving Osseointegration: By adding nanoscale texturing to the surface of bone implants, we create a much more natural interface for bone cells to stick to. This encourages the patient’s own bone to grow directly onto the implant, creating a bond that is stronger, more stable, and lasts longer.
- Reducing Inflammation: Certain nanocoatings can be designed to release anti-inflammatory drugs right where they’re needed. This helps calm the body’s immune response at the implant site, preventing the kind of chronic inflammation that can lead to tissue damage and implant rejection.
By using nanomaterials to create these advanced scaffolds and smarter coatings, regenerative medicine is getting closer to its ultimate goal: helping the body heal itself completely, restoring function, and improving quality of life for millions.
Combining Diagnostics and Therapy with Theranostics
So far, we’ve looked at how nanotechnology can deliver drugs and diagnose diseases separately. But what if you could roll both functions into a single, elegant tool? That’s the entire idea behind theranostics, one of the most exciting frontiers in all of nanomedicine.
The name itself—a mashup of “therapy” and “diagnostics”—tells you everything you need to know. Think of a theranostic agent as a microscopic ‘seek and destroy’ unit. It’s engineered not just to treat a disease but to first find it, confirm its location, and only then unleash its therapeutic payload with pinpoint accuracy.
This two-in-one approach tackles a huge challenge in medicine, especially in cancer treatment. Doctors often have to base their decisions on static scans or biopsies that only capture a single moment in time. Theranostics, on the other hand, gives them a dynamic, real-time window into what’s happening inside the body.
How Does a Single Nanoparticle Do Two Jobs?
The real magic of a theranostic nanoparticle is in its clever, multi-layered design. Scientists can engineer a single particle to carry several different components, each with its own specific job. It’s like building a microscopic Swiss Army knife for fighting disease.
Here’s a quick breakdown of how a typical theranostic agent might work when it’s sent to find a tumor:
- Homing Beacon: The nanoparticle’s surface is decorated with molecules that act like a biological GPS, guiding it directly to cancer cells while letting it slip past healthy tissue.
- Diagnostic Signal: It carries an imaging agent, like a quantum dot or magnetic material. When the particle locks onto the tumor, this agent “lights up” on an MRI or PET scan, showing doctors exactly where the disease is and how big it is.
- Therapeutic Payload: Tucked safely inside is a powerful drug, like a chemotherapy agent. This cargo stays dormant and harmless until it reaches its destination.
- Controlled Release: Once doctors confirm the nanoparticles have gathered in the right spot, they can trigger the drug release using an external signal, like a specific frequency of light or an ultrasound wave.
This process allows for an incredibly controlled and personalized treatment. Doctors can literally see if the drug is building up in the tumor before they activate it, ensuring the therapy is aimed precisely where it’s needed most.
With theranostics, we are moving from a “treat and see” model to a “see and treat” model. This shift promises to take the guesswork out of therapy, allowing doctors to adjust treatments on the fly based on real-time feedback from the patient’s own body.
The Benefits of a Combined Approach
The beauty of theranostics is its efficiency and precision. By combining these steps, it opens the door to some major advantages for both patients and doctors, helping fine-tune treatments in ways that just weren’t possible before.
Some of the key benefits include:
- Truly Personalized Medicine: Treatment is no longer a one-size-fits-all game. Clinicians can verify that a drug is actually reaching its target in a specific patient before committing them to a full, grueling course of therapy.
- Real-Time Monitoring: Doctors can track how well a treatment is working as it happens. They can watch a tumor shrink in response to the drug and adjust the dosage or strategy on the fly.
- Fewer Side Effects: By making sure the drug is only released at the disease site, the exposure to healthy tissues is dramatically lowered. This is a game-changer for minimizing the harsh side effects that come with powerful drugs like chemotherapy.
Theranostics represents a huge leap toward smarter, safer, and more effective medicine. By uniting diagnostics and therapy into one powerful package, this nanotechnology application in medicine is setting the stage for the future of personalized care.
Understanding The Risks And Ethical Questions
As exciting as nanomedicine is, this powerful new toolkit isn’t a magic bullet. It comes with its own set of very real challenges and tough questions. Like any major scientific leap forward, getting from a cool lab result to a safe clinical treatment requires an honest look at the potential downsides. Responsible innovation means tackling these hurdles head-on.
One of the biggest areas of concern is nanotoxicity. What happens to these microscopic particles after they’ve dropped off their drug payload or finished their diagnostic job? Because nanoparticles are tiny enough to slip inside cells and can build up in organs like the liver and spleen, we have to be absolutely sure they don’t cause long-term harm.
This is a surprisingly tricky question. The effects can change dramatically based on a nanoparticle’s size, shape, what it’s made of, and its surface coating. A huge focus right now is designing particles that are fully biodegradable—basically, building them to do their job and then break down into harmless bits the body can easily flush out.
Navigating The Regulatory Maze
Beyond the biological risks, there are major logistical hoops to jump through. Getting any new medical treatment approved is already a long, complicated slog, and nanomedicines get an extra layer of scrutiny. Regulatory bodies are still figuring out how to build new frameworks to properly evaluate the unique safety profile of these sophisticated therapies.
The real balancing act for regulators is creating rules that are strict enough to keep patients safe without completely killing the innovation that could lead to breakthrough treatments. It’s a delicate dance that demands tight collaboration between scientists, companies, and government agencies.
This all involves years of extensive preclinical and clinical trials. Each one is designed to answer critical questions about how these particles behave in the human body over time. It’s a slow, expensive, but non-negotiable step to ensure that any nanomedicine that makes it to market is both safe and effective for everyone.
The Ethical Dimension of Access
Finally, the incredible power of nanomedicine brings up some big ethical questions, with accessibility and equity right at the top of the list. These treatments are the product of years of extremely expensive research and development. That means when they first become available, they will almost certainly be very, very expensive.
This forces us to ask a tough societal question: how do we prevent these amazing therapies from becoming just another tool that widens the gap between the rich and the poor?
- Cost and Insurance: Will healthcare systems and insurance companies be able to cover these advanced treatments? Or will they only be an option for those who can afford to pay out of pocket?
- Global Equity: How do we make sure patients in lower-income countries get a chance to benefit from these life-saving innovations, and not just people in wealthy nations?
- Informed Consent: As these treatments get more complex, how can we be sure that patients truly understand the risks and benefits of these new nanoparticle-based therapies?
Figuring out these issues is every bit as important as solving the scientific puzzles. The end goal isn’t just to invent powerful new medicines, but to ensure they benefit all of humanity. As the field grows up, a relentless focus on safety, rigorous testing, and fair access will be the only way to responsibly guide this technology into the future.
Got Questions About Nanomedicine?
As we dig into the incredible world of medical nanotechnology, it’s totally normal for questions to pop up. This field can sound like something straight out of science fiction, but a lot of these innovations are closer to your local clinic than you might imagine. Let’s tackle some of the most common questions to get a clearer picture of how nanomedicine works and the difference it’s making today.
Is Nanomedicine Actually Used in Hospitals Today?
Yes, absolutely. While it might sound futuristic, several nanomedicines are already a standard part of patient care. The most common examples are found in cancer treatment, where drugs like Doxil® and Abraxane® have been in an oncologist’s toolkit for years.
These treatments work by tucking powerful chemotherapy agents inside tiny nanoparticles. It’s a simple yet brilliant strategy that helps the medicine zero in on tumors more effectively while sparing the rest of the body from the harshest side effects. Beyond cancer, nanoparticles are also used in certain medical imaging agents to help doctors get sharper MRI or CT scans, and they’re the key ingredient in many of the rapid diagnostic tests you might find at a clinic.
What Makes Nanoparticles So Good for Delivering Drugs?
The magic is in the precision. Think about it: traditional drugs often flood the entire body, hitting healthy cells just as hard as the diseased ones. This is exactly why treatments like chemotherapy can be so grueling for patients. Nanoparticles, on the other hand, act more like a tiny, GPS-guided delivery service.
Scientists can engineer the surface of a nanoparticle, attaching specific molecules that act like a key to a lock. These molecules are designed to fit only the receptors found on diseased cells, like cancer cells.
This targeting makes sure the drug payload gets released right where the problem is. Not only does this make the treatment more powerful, but it also drastically cuts down on the collateral damage to healthy tissue, which can make a world of difference for a patient’s quality of life.
Are Nanoparticles Safe to Put in The Human Body?
Patient safety is, without a doubt, the number one priority and the focus of a massive amount of ongoing research. The main concern is making sure these nanoparticles don’t hang around in the body and cause problems down the line—a field of study known as nanotoxicity.
Researchers spend countless hours studying how nanoparticles of different sizes, shapes, and materials are handled by the body. They track how they’re processed, broken down, and eventually cleared out by our natural systems. A huge goal in the field is to design particles that are fully biodegradable, meaning they do their job and then simply dissolve into harmless bits that the body can easily flush out.
Before any nanomedicine ever makes it to a patient, it has to pass through an incredibly demanding series of safety and efficacy trials set by regulatory bodies like the FDA. These strict protocols are there to confirm that the benefits for the patient massively outweigh any potential risks. It’s a rigorous process that ensures every piece of approved nanotechnology in medicine meets the highest possible standards of care.
At maxijournal.com, we dive deep into the science and technology shaping our future. Explore more clear, accessible articles on everything from health to business. Discover your next great read at maxijournal.com.
Discover more from Maxi Journal
Subscribe to get the latest posts sent to your email.