At its core, bioinformatics is a blend of biology, computer science, and statistics. It’s the toolkit we use to make sense of the overwhelming amount of data generated by modern biological research. Think of it as the discipline dedicated to decoding life’s instruction manual—DNA, RNA, and proteins—and turning that raw code into incredible scientific discoveries.
Decoding Life’s Instruction Manual

Imagine trying to read every single book in the world’s largest library, all at once, just to find one specific word. That’s pretty close to the scale of the challenge biologists face. The human genome alone contains over 3 billion letters of code. It’s a staggering amount of information, far too vast for any person to process on their own.
This is exactly where bioinformatics comes in. It provides the heavy-duty computational power needed to manage, analyze, and pull real knowledge from this biological “big data.” So, instead of working with pipettes and test tubes, a bioinformatician’s lab is full of algorithms, databases, and powerful computers.
The Three Core Pillars
Bioinformatics isn’t just one subject; it’s a true interdisciplinary field that gets its power from three key areas working together:
- Biology: This is the foundation. It gives us the fundamental questions we want to answer and the raw material to work with, like DNA sequences, protein structures, and data on which genes are active.
- Computer Science: This provides the muscle. It brings the algorithms, software development, and database management needed to handle massive datasets without breaking a sweat.
- Statistics: This is the brain of the operation. It offers the mathematical frameworks to tell the difference between a meaningful biological signal and just random noise, making sure our conclusions are solid.
In essence, bioinformatics acts as the crucial bridge connecting the complex world of modern biology with the raw power of computation. It translates the intricate language of life into a format that computers can understand, revealing patterns that would otherwise be completely invisible to us.
This powerful combination is what lets scientists tackle some of biology’s biggest puzzles, from pinpointing the genes responsible for a disease to tracing a species’ evolutionary family tree. Below is a quick breakdown of how these disciplines fit together.
The Core Pillars of Bioinformatics
This table shows how each discipline contributes to making bioinformatics a reality.
| Discipline | Role in Bioinformatics |
|---|---|
| Biology | Provides the raw data (e.g., DNA, RNA, proteins) and the biological context needed for the analysis. |
| Computer Science | Develops the algorithms, software, and databases required to store, manage, and process the data. |
| Statistics | Creates the mathematical models and tests to find significant patterns and validate scientific findings. |
By weaving these fields together, bioinformatics elevates biological research from simple data collection into the exciting realm of true interpretation and groundbreaking discovery.
To really get a feel for why bioinformatics is such a big deal today, it helps to rewind the clock a bit. Before powerful computers were standard lab equipment, analyzing biological data was a slow, manual, and often painful process. Scientists were hitting a wall: they could generate new data way faster than they could possibly make sense of it. This data pile-up created a bottleneck that only computers could break through.
The story of bioinformatics doesn’t start with silicon chips, though. It starts with the fundamental human drive to understand the very machinery of life. Back in the mid-20th century, researchers were making incredible discoveries about proteins—the tiny molecular engines that do just about everything in our cells. The real breakthrough came when they started figuring out how to map their exact structures.
The Dawn of Sequence Analysis
This new age of biological data truly kicked off in 1955. That’s when Frederick Sanger announced he had mapped the full amino acid sequence of bovine insulin. This was a monumental achievement, earning him a Nobel Prize and marking the first time anyone had fully decoded a protein. In those early days, comparing two sequences meant writing them out on long strips of paper and literally sliding them past each other to spot similarities. It was a tedious, eye-straining method that massively limited what researchers could accomplish. You can explore a deeper dive into these early milestones and their impact.
As more and more sequence data trickled in, it became painfully obvious that these manual methods just weren’t going to cut it. Biology needed a better, more mathematical way to compare sequences.
The problem wasn’t a lack of information; it was a lack of tools powerful enough to interpret it. Biologists were building massive libraries of genetic text but had no search engine to navigate them.
This desperate need for computational horsepower is what gave birth to bioinformatics as a real discipline. The solution came from the world of computer science, with brilliant minds developing algorithms specifically for biological puzzles.
From Manual Labor to Computational Power
The field took a massive leap forward in 1970 with the Needleman-Wunsch algorithm. This was the first dynamic programming method that could find the optimal alignment between two sequences. Suddenly, computers could measure biological similarity with mathematical rigor. Scientists were no longer just eyeballing alignments; they could generate objective scores to back up their hypotheses.
This computational breakthrough happened at the perfect time. A few other key developments were setting the stage for modern genetics and creating an even bigger demand for data management:
- Recombinant DNA: In 1972, Paul Berg created the first-ever recombinant DNA molecule. This opened the door to genetic engineering and gave scientists the power to directly manipulate DNA sequences.
- Public Databases: The Brookhaven Protein Data Bank launched in 1973, becoming the first open-access library for 3D protein structures. It started with just seven entries but set a vital precedent: biological data should be shared publicly for everyone’s benefit.
These innovations unleashed a tidal wave of new information. As sequencing proteins and DNA got faster and cheaper, the sheer volume of data made computers an absolute necessity. Bioinformatics went from being a niche academic curiosity to the essential engine driving modern science, paving the way for massive efforts like the Human Genome Project.
Core Concepts And Common Methods Explained
To really get what bioinformatics is all about, you have to look under the hood. It’s a field built on a few foundational ideas and a toolkit of digital methods that bioinformaticians use every day to turn the raw language of biology into something we can actually understand and use. Let’s build this up piece by piece, starting with the most basic task of all: reading the letters of life.
At its very core, a huge chunk of bioinformatics is about sequences. These are the long strings of letters—A, T, C, G for DNA, or the 20 amino acids for proteins—that write the instructions for all living things. The first job is usually sequence analysis, which is just a broad term for all the ways we try to decode the information hiding in these biological strings. This can be anything from spotting where a gene begins and ends to predicting what a specific protein actually does.
Reading and Comparing Biological Text
One of the most powerful and common methods is sequence alignment. The best way to think about it is like using the “Compare Documents” feature in a word processor. You take two sequences—say, the same gene from a human and a mouse—and you line them up to see where they match and where they’re different.
These similarities and differences are incredibly revealing. They can point to a shared evolutionary history, suggesting both genes came from a common ancestor millions of years ago. They can also hint at function. If you see a specific part of a gene that is almost identical across hundreds of species, that’s a massive clue that this region is absolutely critical for survival.
This whole process is driven by some pretty smart algorithms that score the alignments, giving points for matches and taking them away for gaps or mismatches. The most famous tool for this, by far, is BLAST.
BLAST (Basic Local Alignment Search Tool) is basically the search engine for biology. A scientist can discover a new gene, pop its sequence into BLAST, and instantly scan gigantic global databases for anything that looks similar. In a matter of seconds, this simple search can offer a flood of clues about the gene’s possible function and where it came from on the evolutionary tree.
Understanding The Bigger Picture: Genomics And Proteomics
While sequence analysis can zoom in on a single gene or protein, bioinformatics also operates on a much, much grander scale. This is where we get into the massive fields of genomics and proteomics.
- Genomics is the study of an organism’s entire library of genes—its genome. Instead of looking at one gene at a time, genomicists analyze all of them at once to figure out how they work together as a complex system to run the show. This means mapping where genes are, finding tiny variations between individuals, and studying the overall architecture of our DNA.
- Proteomics does the exact same thing, but for proteins—the proteome. Proteins are the little machines that do most of the actual work in our cells. Proteomics tries to figure out which proteins are present in a cell, how many there are, and how they all interact with each other.
Both of these “omics” fields produce an absolutely staggering amount of data. It’s so much that they are completely dependent on bioinformatic tools to make any sense of it. The skills for managing and interpreting this data have a lot in common with another exploding field. If this is sparking your interest, you might also want to read up on what data science is in our complete explainer for beginners.
Key Bioinformatic Workflows And Databases
Bioinformaticians don’t work in isolation. They rely on well-trodden workflows and massive public databases that act as a shared library for the entire global scientific community. A typical project isn’t about using one magic tool; it’s about stringing together several different tools to answer a single question.
For instance, a classic task is figuring out the evolutionary tree for a group of species using a single, shared gene. That workflow would look something like this:
- Data Retrieval: First, the researcher would grab the sequences for that gene from all the different species. They’d almost certainly get them from a public database like GenBank, which is home to trillions of DNA bases.
- Multiple Sequence Alignment: Next, they’d use a tool to line up all of those sequences at the same time. This highlights the parts of the gene that have been conserved across the whole group over millions of years.
- Phylogenetic Analysis: Finally, they feed that alignment data into another program to build a phylogenetic tree. This is a branching diagram that visually lays out the evolutionary relationships, just like a family tree.
This ability to build on a global collection of shared data is what makes modern biology move so fast. Databases like GenBank for sequences and the Protein Data Bank (PDB) for 3D protein structures are the bedrock of the field. Without these computational methods and shared resources, trying to understand the full complexity of biology would be a truly impossible task.
How Bioinformatics Is Transforming Our World
Alright, we’ve walked through the core methods, but this is where the rubber really meets the road. All that abstract stuff—sequence alignment, genomics, algorithms—translates into tangible breakthroughs that are actively changing medicine, farming, and our deepest understanding of life itself. The impact isn’t just academic; it’s being felt in major industries, helping us solve some of the world’s biggest problems.
The sheer explosion of biological data is what’s fueling all this. Consider GenBank, the public sequence database. It kicked off in 1982 with about 680,000 DNA bases. By 2023, it held over 10 trillion. That kind of exponential growth has created a massive need for the tools to make sense of it all. As a result, the bioinformatics market hit $14.5 billion in 2023 and is on track to reach $40 billion by 2030, which tells you just how vital this field has become. You can get a great sense of the timeline of these groundbreaking genetic discoveries that paved the way for today’s progress.
Paving The Way For Personalized Medicine
Maybe the most profound change we’re seeing is in healthcare, with the shift toward personalized medicine. The old “one-size-fits-all” model for treating diseases is slowly giving way to therapies designed for a person’s unique genetic code.
Think about two patients with the same type of cancer. Years ago, they’d likely get the exact same chemotherapy, but with wildly different results. Now, bioinformaticians can analyze a tumor’s DNA to pinpoint the specific mutations making it tick. This genetic blueprint lets doctors choose a drug that targets that exact mutation, leading to much better outcomes with fewer nasty side effects.
This isn’t just theory; it’s happening right now. Big initiatives like the UK’s 100,000 Genomes Project proved it works, successfully diagnosing 25% of participants who had rare diseases and helping guide treatment for most of the cancer patients involved.
This infographic gives a quick look at some of the core methods driving these breakthroughs.
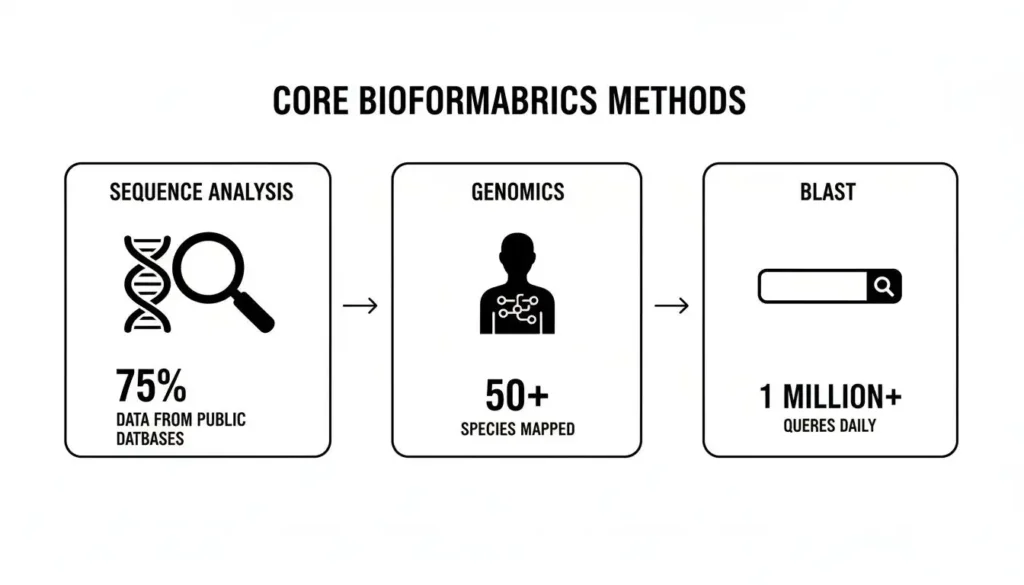
From reading individual DNA strands with sequence analysis to using powerful search tools like BLAST, these techniques are the bedrock of modern biological discovery.
Accelerating Drug Discovery And Development
Getting a new drug from the lab to the pharmacy is a ridiculously long and expensive journey—we’re talking over a decade and billions of dollars. Bioinformatics is completely flipping the script, making drug discovery smarter, faster, and cheaper.
Instead of the old-school method of testing thousands of chemical compounds in a wet lab, scientists can now build computational models to predict how a drug might interact with a target protein in the body. This in silico (meaning computer-simulated) approach weeds out the duds and highlights the most promising candidates before a single physical experiment has to be run.
By simulating biological interactions on a computer, researchers can fail faster and cheaper. This allows them to focus resources on the drug candidates with the highest probability of success, a critical advantage in a high-stakes industry.
We saw this computational power in action during the COVID-19 pandemic. Scientists across the globe sequenced and shared over 1.5 million SARS-CoV-2 genomes through databases like GISAID. This massive, collaborative data dump, made possible by bioinformatics, was the key to developing highly effective vaccines in record time.
Beyond Human Health: New Frontiers
The reach of bioinformatics extends far beyond the doctor’s office. The very same principles are being put to work to tackle huge global challenges in other fields.
- Agriculture: Scientists are using genomics to engineer crops that can better withstand drought, pests, and disease. By zeroing in on the genes for these traits, they can breed more resilient and productive plants to help feed a growing world.
- Environmental Science: Out in the field, researchers are analyzing DNA from microorganisms in soil and seawater to understand entire ecosystems. This is called metagenomics, and it helps us monitor the health of our planet and even find new enzymes that can be used to clean up pollution.
- Evolutionary Biology: Bioinformatics lets us compare the genomes of different species to map out the tree of life with incredible accuracy. It helps us piece together how life evolved and how everything is connected, answering some of the most fundamental questions about where we came from.
From designing life-saving therapies to growing hardier crops, bioinformatics is the invisible engine powering countless scientific advances. It’s the critical link between the microscopic world of DNA and the biggest challenges we face every day.
The Human Genome Project: A Case Study

If you really want to see bioinformatics in action, there’s no better example than its biggest success story: the Human Genome Project (HGP). When it kicked off in 1990, it felt like biology’s version of a moonshot. The goal was ridiculously ambitious: read and map every single letter in our genetic instruction book—all 3.1 billion of them.
This wasn’t just a big science project; it was something that would have been physically impossible without a deep partnership between biology and computer science. The challenge was staggering. Back then, DNA sequencing technology could only read tiny, fragmented pieces of the genome at a time. The job was like taking thousands of encyclopedias, running them through a shredder, and then trying to tape all the tiny strips back together in the correct order.
The Ultimate Jigsaw Puzzle
This is where bioinformatics stole the show. Scientists had to invent incredibly sophisticated algorithms just to tackle this massive assembly problem. These computational tools were the expert puzzle solvers, scanning millions of DNA fragments, looking for the slightest overlaps, and then methodically stitching them together into long, unbroken stretches of genetic code.
Without that computational heavy lifting, the project would have been dead in the water. The sheer volume of data was far too much for any team of humans to analyze.
Think about this: if you printed out the human genome, it would fill around 200 million pages of text. Trying to piece that together by hand would have taken centuries, not just a few years. Bioinformatics was the only way to make it happen in a human lifetime.
The HGP became a masterclass in international scientific teamwork. It was a global effort, with 20 institutions across six countries, all spearheaded by the US Department of Energy and the National Institutes of Health. With a $3 billion investment, it pushed science to its limits. Thanks to the power of bioinformatics, the project finished ahead of schedule, with the complete genome sequence announced in 2003. This achievement blew open countless new doors for research. You can dig deeper into its impact on modern biology and its economic legacy if you’re interested.
Creating a Legacy of Open Data
One of the project’s most powerful ideas was making all the data immediately and freely available. The 1996 Bermuda Principles established a rule that all new sequence data had to be uploaded to public databases within 24 hours. This open-access philosophy meant that scientists anywhere in the world could jump in and start working with the data, which massively accelerated the pace of discovery.
This shared resource became the bedrock for thousands of studies into human health. Researchers could finally pinpoint specific genes linked to diseases like cystic fibrosis or Huntington’s, paving the way for better diagnostic tests and potential treatments. This work was a direct catalyst for fields like gene therapy. If you’re curious about that, we have a great guide on what gene therapy is and how it works.
But the project’s most enduring legacy might be how it turbocharged sequencing technology itself. The massive scale of the HGP created a demand that drove innovation at a breakneck pace, causing the cost of sequencing to plummet. A process that once cost millions of dollars per genome can now be done for just a few hundred. This democratization of genomics is what kicked off the modern era of personalized medicine, turning a once-in-a-generation scientific effort into a routine part of today’s healthcare.
The Future of Bioinformatics
So, where is this field that decodes life’s instruction manual headed? To put it simply, the future of bioinformatics is unfolding at a dizzying pace, largely thanks to the growing roles of Artificial Intelligence (AI) and Machine Learning (ML). These aren’t just buzzwords; they’re fundamentally changing how we approach biological data.
We’re moving past simply finding existing patterns. AI models can now predict biological outcomes with a level of accuracy that was once science fiction. This means we can forecast a person’s risk for a certain disease just by looking at their genome. Or we can design entirely new drugs inside a computer—a process known as in silico drug discovery—slashing the time it takes to develop new medicines.
New Frontiers in Biological Exploration
The field is also pushing into brand-new territory. One of the most exciting areas is metagenomics, which takes us from studying a single organism to analyzing the genetic material of entire microbial communities all at once. Think about being able to sequence every strand of DNA in a scoop of soil or a drop of seawater.
This gives us an unprecedented view of an ecosystem’s biodiversity and function, and the implications are huge for everything from environmental science to human health. By truly understanding our gut microbiome—the trillions of bacteria living inside us—we’re unlocking novel ways to treat a whole host of conditions.
As we generate more complex biological data, the real challenge shifts from collection to interpretation. The future belongs to those who can build the smartest tools to find the signal in the noise, turning massive datasets into actionable knowledge.
Navigating Challenges and Responsibilities
Of course, this exciting future isn’t without its hurdles. As genomic data becomes more and more common, the ethical questions surrounding its use are getting louder and more urgent. Genomic data privacy is probably the biggest challenge on the horizon. How do we protect the most personal information a person has while still using it to push science forward?
Getting that balance right between progress and privacy is crucial for keeping the public’s trust. The field needs to double down on developing rock-solid security protocols and clear ethical guidelines to make sure this powerful information is always handled responsibly.
Despite these challenges, the outlook is incredibly bright. Bioinformatics is set to remain a central pillar of modern science, acting as the computational engine we need to tackle some of humanity’s biggest problems. From fighting pandemics to securing our food supply, the ability to read and understand biological data will be one of our most vital tools for decades to come.
Frequently Asked Questions About Bioinformatics
As you get your feet wet in bioinformatics, a few questions always seem to pop up. Here are some straightforward answers to help you connect the dots and figure out what it really takes to succeed in this field.
Do I Need To Be A Programmer To Work In Bioinformatics?
The short answer? Not always, but it’s a massive advantage. While some roles might lean more toward the biology side of things, most core bioinformatics research is built on programming.
You’ll find that languages like Python or R are the bread and butter for analyzing data, creating custom tools, and automating repetitive tasks. Plenty of bioinformaticians start as biologists and pick up coding along the way (or vice versa). The real key is learning to speak both “languages” fluently.
What Is The Difference Between Bioinformatics And Computational Biology?
You’ll often hear these terms used interchangeably, and there’s a lot of overlap, but there is a subtle difference in focus. Here’s a simple way to think about it:
- Bioinformatics is generally about developing and applying the tools—the software, algorithms, and databases—needed to manage and interpret biological data. It’s often very hands-on and practical.
- Computational Biology is more about using that data to build theoretical models and run simulations of biological systems. It aims to answer fundamental questions about how life works using quantitative approaches.
In a nutshell, bioinformatics builds the workbench and the tools, while computational biology uses them to model and test hypotheses about the machinery of life.
How Can I Start Learning Bioinformatics?
Jumping in is easier than ever. A great starting point is checking out online courses on platforms like Coursera or edX, which have structured programs from top universities.
If you prefer learning by doing, a fantastic free resource is Rosalind. It’s a platform that teaches you bioinformatics concepts by having you solve programming puzzles. No matter which path you choose, building a solid foundation in both biology and a programming language is the most crucial first step.
For those planning to dive into academic research, understanding the structure of scientific work is also incredibly helpful. Our guide on how to write a research paper without the overwhelm can give you a peek into the field’s methodology.
What Is The Biggest Ethical Challenge In Bioinformatics?
Without a doubt, data privacy stands out as a massive ethical hurdle. Your genomic data is the most personal information you have, revealing everything from your ancestry and health risks to potential future medical issues.
The biggest challenge is ensuring this data is stored securely, used responsibly, and shielded from misuse. Preventing things like genetic discrimination from employers or insurance companies is a critical responsibility the field has to get right as it becomes more and more a part of our daily lives.
At maxijournal.com, we publish daily insights at the intersection of science, technology, and more. Explore fresh perspectives by visiting us at https://maxijournal.com.
Discover more from Maxi Journal
Subscribe to get the latest posts sent to your email.


Pingback: Immunotherapy for Cancer: Key Facts and Insights Detailed
Pingback: New Hair Growth Solutions: Uncovering Effective Options
Pingback: Exploring CRISPR: How Gene Editing Works in Simple Terms