The primordial soup theory is one of the most famous ideas about how life got started. It paints a picture of Earth’s ancient oceans as a rich broth of chemicals that, sparked by lightning and fierce sunlight, cooked up the very first building blocks of biology. This is the classic story of how life could have emerged from non-living matter billions of years ago.
Decoding the Primordial Soup Hypothesis

To really get your head around what is the primordial soup, you have to imagine an Earth that would be completely alien to us. Forget blue skies and clear oceans. We’re talking about a planet around 3.7 to 4.0 billion years ago with a hazy, reddish sky, a landscape dominated by volcanoes, and murky, brownish seas. This was the stage.
The central concept, known as the Oparin-Haldane hypothesis, was pieced together separately by Aleksandr Oparin and J.B.S. Haldane back in the 1920s. They both envisioned that Earth’s early atmosphere was “reducing”—a chemical term meaning it had virtually no free oxygen. Instead, it was thick with simpler gases.
The Key Ingredients
This ancient atmosphere was like a planetary-scale kitchen stocked with a few basic, but powerful, ingredients. To understand how the “soup” could have formed, you need to consider the essential components Oparin and Haldane proposed.
The table below breaks down the three main elements that made this chemical experiment possible.
| Key Components of the Primordial Soup Hypothesis |
| :— | :— | :— |
| Component | Description | Example |
| Reducing Atmosphere | An atmosphere lacking free oxygen, rich in simple molecules that could easily donate electrons to form new compounds. | Methane (CH₄), ammonia (NH₃), hydrogen (H₂), water vapor (H₂O). |
| Energy Sources | Intense and constant energy was needed to break the chemical bonds of the simple atmospheric gases. | Frequent lightning strikes, intense solar ultraviolet (UV) radiation, and volcanic heat. |
| Liquid Water | The planet’s oceans served as a vast container where these chemicals could dissolve, mix, and react. | Warm, shallow pools or vast stretches of the early ocean. |
These three components worked together, turning the early Earth into a massive, self-running chemistry lab. The stage was set for something incredible to happen.
From Simple Gases to Life’s Building Blocks
The core of the hypothesis is that life wasn’t a single, magical event. It was the result of chemical complexity slowly building up over an immense amount of time.
The Chemical Cradle of Life: The primordial soup hypothesis sees the ocean as more than just water; it was an active chemical reactor. This dynamic environment allowed the building blocks of life to gather, concentrate, and begin the long, slow march toward the first cells.
As J.B.S. Haldane explained in his seminal 1929 paper, the lack of oxygen was crucial. Without it, any new organic molecules that formed wouldn’t be immediately destroyed by oxidation. They could stick around. You can explore more about Haldane’s foundational concepts on the origin of life.
Over millions of years, these compounds—like amino acids (the parts of proteins) and nucleotides (the parts of DNA and RNA)—would have collected in the warm, shallow waters, creating the nutrient-rich broth we now call the primordial soup.
This powerful idea set the stage for one of the most famous experiments in science, which aimed to prove that this transformation from simple chemistry to the building blocks of life was not just possible, but plausible.
Recreating Ancient Earth in a Modern Lab

The idea of a primordial soup was compelling, but for decades, it was just an idea. How could anyone test a hypothesis about chemistry on a planet billions of years in the past? In 1953, a graduate student named Stanley Miller, under the guidance of his Nobel laureate advisor Harold Urey, decided to find out.
Their approach was brilliant in its simplicity: they would build a closed-system version of early Earth right there in the lab. The now-famous Miller-Urey experiment aimed to see if life’s basic ingredients could form from simple chemistry, given the conditions Oparin and Haldane had proposed. It was a bold attempt to cook up a primordial soup from scratch.
Designing a Miniature Planet
To pull this off, Miller and Urey built a self-contained loop of glass flasks and tubes. Each piece of the apparatus was designed to represent a key feature of the ancient world.
- The “Ocean”: A flask of water was gently heated to produce water vapor, simulating the warm, early oceans.
- The “Atmosphere”: This vapor then moved into a larger flask filled with the gases believed to make up Earth’s early air: methane (CH₄), ammonia (NH₃), and hydrogen (H₂).
- The “Lightning”: Inside the “atmosphere” flask, electrodes sparked continuously, mimicking the violent, constant lightning storms of a young planet.
- The “Rain”: Finally, a condenser cooled the gases, causing the water vapor—along with any new chemicals it had picked up—to condense and fall back into the “ocean,” just like rain.
They let this cycle run nonstop for a week. The goal was to just let the simple ingredients “cook” under a constant blast of energy and see what happened. Slowly, the clear water in the ocean flask turned a murky brown, and then a deep red. Something was definitely happening.
After just one week, the results were stunning. An analysis of the water revealed that the experiment had spontaneously created organic molecules, including several different amino acids—the essential building blocks of proteins.
This was a massive breakthrough. It was the first hard, experimental evidence that the raw materials for life could actually emerge from simple, non-living chemistry. The primordial soup wasn’t just a fantasy anymore; it was a plausible scientific scenario. Miller’s work showed that the jump from a sterile planet to one filled with life’s ingredients might not have been as difficult as everyone thought.
A Discovery More Successful Than Imagined
For decades, that’s where the story stood. Miller’s original 1953 analysis had identified five different amino acids in his chemical brew—a landmark success that completely changed our view of abiogenesis, the origin of life from non-living matter. But the true scope of his success wouldn’t be known until much later.
Years later, scientists got their hands on samples from Miller’s experiments that had been sealed and preserved since 1958. Using modern, highly sensitive analytical equipment, they re-examined the contents and found that his primordial soup was far richer than he ever knew. His experiments had actually produced a staggering 23 different types of amino acids, a yield far beyond what the technology of his time could detect.
This later analysis showed Miller’s setup had generated roughly 4.6 times more distinct organic compounds than he had originally reported. You can read more details about these later findings on the Miller-Urey experiment. This posthumous discovery added incredible weight to the original findings, demonstrating that the chemical reactions on early Earth were likely even more robust and creative than first imagined.
The Chemical Recipe That Sparked Life
So, the Miller-Urey experiment showed us that the basic parts for life could just form spontaneously. But what were these parts, and how did they actually link up to build something more complex? The best way to think about the primordial soup isn’t as a random sludge, but as a perfectly stocked pantry for life’s very first recipe.
To get a handle on how life might have kicked off, we have to meet the key molecular players. These are the fundamental units that, even today, make up every living thing on our planet. Without them, that leap from simple chemistry to complex biology just wouldn’t have happened.
You can think of this recipe as needing a few different kinds of molecular “LEGO bricks.” Each one has a specific job, and when you put them all together, they build the intricate machinery we call life.
Life’s Fundamental Building Blocks
Three main families of organic molecules were absolutely crucial for getting things going in that primordial soup. They weren’t just floating around randomly; they were the raw materials, waiting for a blueprint.
- Amino Acids: These are the building blocks of proteins. Think of them like super versatile LEGO bricks you can snap together into long chains. These chains build everything from structural supports (like collagen) to tiny machines (enzymes) that make chemical reactions happen.
- Nucleotides: These are what make up DNA and RNA. Picture them as the letters of an alphabet, used to write life’s instruction manual. They’re the key to storing genetic information and passing it on.
- Lipids (Fats): These are molecules that just don’t mix with water. Their most important role was forming the very first cell membranes. Lipids are like the walls of a LEGO house, creating a self-contained little bubble that separates the chemistry on the inside from the chaos on the outside.
These three groups gave you the structure, the instructions, and the container. But just having a pile of bricks isn’t enough to build a house; you need energy and a builder to put it all together.
The real beauty of the primordial soup idea is that it explains where both the ingredients and the assembly process came from. The same chaotic forces that defined early Earth also supplied the raw energy needed to join these simple molecules into something far more complex and useful.
The Energy That Built Complexity
Early Earth was an incredibly violent place. But it turns out, all that violence provided exactly the kind of energy needed to push prebiotic chemistry forward. The simple molecules in the “soup” didn’t just casually bump into each other; they were actively slammed together by powerful environmental forces.
The main energy sources were pretty intense:
- Intense Ultraviolet (UV) Radiation: With no ozone layer to shield the planet, the sun’s harsh UV rays hammered the surface, breaking old chemical bonds and sparking new ones.
- Constant Lightning Strikes: The atmosphere was thick with electrical storms. Every single lightning bolt delivered a massive jolt of energy that was perfect for forging new organic compounds.
- Volcanic Heat: Widespread volcanic activity and deep-sea hydrothermal vents acted like chemical cooktops, providing sustained heat that sped up reactions.
This constant blast of energy was the “hand” that started snapping the molecular bricks together. It drove a critical process that really marks the beginning of biological complexity: polymerization.
From Single Molecules to Long Chains
Polymerization is just the chemical term for linking single molecules (monomers) together to form long, repeating chains (polymers). This was the make-or-break step that turned the simple primordial soup into something much more interesting. A single amino acid can’t do much on its own, but a chain of them—a protein—can fold into a specific shape and actually do a job.
Imagine you have a bunch of individual beads. Those are the monomers, like amino acids or nucleotides. When you tie them together on a string, the finished necklace is the polymer—a protein or a strand of RNA. This process took the simple stuff made in the Miller-Urey experiment and built it up into the first real macromolecules.
This is one of the most important steps in the origin of life—the move from a loose collection of monomers to structured polymers. It’s the bridge that connects random chemistry to purposeful biology, turning the simple broth of the primordial soup into a workshop where the first hints of life could finally take shape.
How Volcanoes and Meteorites Spiced the Soup
The classic image of a single, planet-wide chemical ocean is a good starting point, but the real story was probably a lot more exciting. Instead of a uniform broth, early Earth likely sizzled with localized “hotspots”—pockets of intense chemical activity running on an accelerated schedule. Think of volcanic regions as the real factories where life’s first ingredients were being mass-produced.
It’s like dropping concentrated flavor packets into a giant pot of water. Rather than diluting everything across a vast ocean, nature created powerful, localized kitchens where conditions were perfect for complex molecules to form. This reframes our understanding of what is the primordial soup from a simple sea to a patchwork of potent chemical engines.
Volcanoes were perfectly suited for the job. They belched out gases that created unique micro-atmospheres, little pockets of air that were radically different from the rest of the planet.
Volcanic Vents as Chemical Factories
Later versions of the famous Miller-Urey experiment dug into this very idea. Scientists started adding gases like hydrogen sulfide (H₂S)—that familiar rotten-egg smell common in volcanic emissions—to the original methane and ammonia mix. The results were stunning.
When this sulfur-rich gas was thrown into the spark chamber, the experiments produced a far richer and more abundant variety of amino acids. This was a huge clue, suggesting a direct link between volcanic activity and the creation of life’s building blocks.
The big takeaway? You didn’t need the entire planet to have a specific atmosphere. All you needed were pockets of intense chemical action around volcanoes. These could have generated a steady supply of organic molecules that then washed into nearby pools and oceans, solving the “concentration problem”—the nagging issue that useful molecules would be too spread out in a global ocean to ever meet and react.
The volcanic model suggests that instead of one giant soup, early Earth was more like a planet of countless “boutique” chemical kitchens, each cooking up a unique menu of organic compounds near vents and geysers.
An Unexpected Connection from Space
This idea of localized chemical factories gets even more interesting when we look up at the sky. The recipe for life wasn’t just being cooked up on Earth; it was also being delivered from space by meteorites. For billions of years, our planet has been pelted by space rocks, and some of them carry some very interesting cargo.
When scientists analyzed the amino acids created in those lab experiments laced with hydrogen sulfide, they noticed something incredible. The types and ratios of the molecules were an almost perfect match for the amino acids found inside carbon-rich meteorites, like the famous Murchison meteorite. As you can learn more from this analysis of primordial soup experiments, this wasn’t a coincidence. The volcanic gases Miller used didn’t mimic the whole ancient atmosphere, but they were a dead ringer for the air around a volcano.
This points to a richer, dual-source model for the primordial soup:
- Terrestrial Production: Earth’s own volcanoes were churning out amino acids in geochemically active hotspots.
- Extraterrestrial Delivery: At the same time, meteorites were delivering a nearly identical set of pre-made ingredients straight from the cosmos.
So, the primordial soup wasn’t just homegrown. It was constantly being “spiced” with deliveries from beyond our world, seriously boosting the planet’s chemical inventory and tipping the odds in favor of life.
Exploring Modern Theories Beyond the Soup
While the classic “primordial soup” idea is a great starting point, it’s definitely not the only game in town. Scientists today have moved beyond just picturing a warm, shallow pond and are exploring some pretty extreme environments where life’s first spark could have appeared. These newer theories aren’t trying to completely replace the old one; think of them as adding new, crucial pieces to a massive puzzle.
A big hang-up with the original soup theory is dilution. Many scientists argue that a planet-wide ocean of chemicals would be far too watery for anything interesting to happen. To build complex molecules, you need your ingredients concentrated in one place with a steady power source. This has pushed the search from open water to more contained, high-energy hotspots.
The Hydrothermal Vent Theory
One of the most exciting alternatives takes us deep into the dark, crushing pressure of the ocean floor. The Hydrothermal Vent Theory proposes that life didn’t start at the sunny surface, but around underwater geysers. These vents are cracks in the seafloor that blast out superheated, mineral-packed water from deep inside the Earth.
This creates a wild environment with what scientists call steep chemical and temperature gradients. Basically, you have a massive difference in heat and chemistry between the scorching vent water and the frigid ocean surrounding it. These gradients act like natural engines, providing the continuous energy needed to kickstart the reactions that form complex organic molecules.
What’s more, these vents are loaded with chemicals like hydrogen sulfide and iron minerals. These can act as catalysts, speeding up the creation of amino acids and other building blocks of life. In essence, this theory moves the primordial kitchen from the surface to a protected, energy-rich spot deep beneath the waves.
The Elegant RNA World Hypothesis
Another major theory tackles a completely different question: the ultimate chicken-and-egg problem. What came first, the genetic instructions (DNA) or the protein machinery that reads them? The RNA World Hypothesis offers a brilliant way out by suggesting that an earlier molecule, RNA, did both jobs.
In our cells today, DNA holds the blueprint, and proteins do the heavy lifting, like catalyzing reactions. You can’t have one without the other—you need proteins to build and read DNA, but you need DNA to make proteins. It’s a paradox.
RNA, however, is a bit of a multi-tool. It can store genetic information just like DNA, but certain types of RNA, called ribozymes, can also fold up and act like protein enzymes to drive chemical reactions. This dual role makes RNA the perfect candidate for the very first self-replicating molecule. The idea is that an “RNA World” got things started before eventually handing the job over to the more stable DNA and more versatile protein system we see today. If you’re interested in how we manipulate these codes now, you can check out our guide on how CRISPR works.
This infographic gives a great overview of how the raw materials for life might have been delivered.
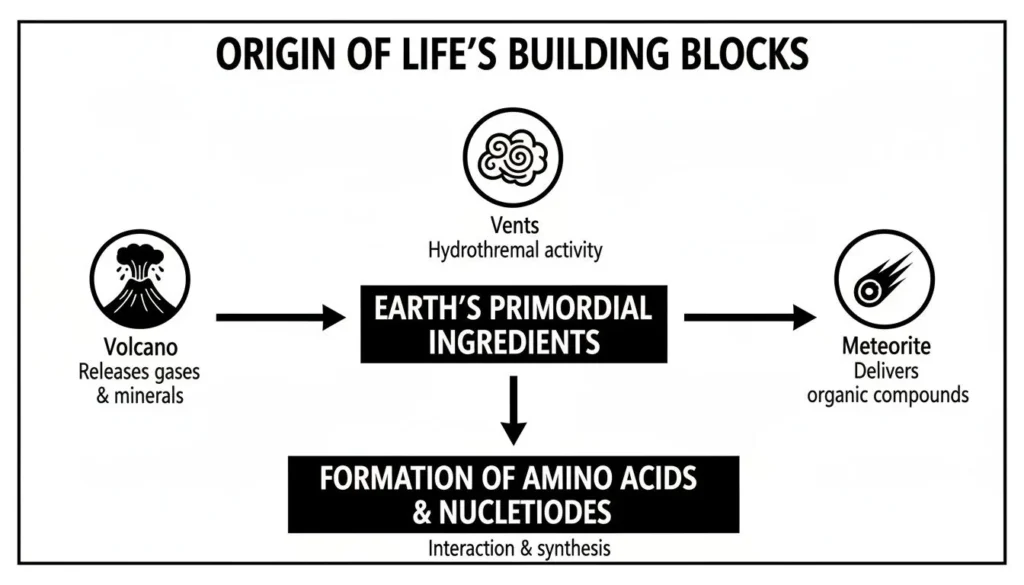
Whether they were cooked up by volcanoes, spewed from deep-sea vents, or delivered from space on meteorites, it seems Earth had several ways to get the building blocks it needed.
Comparing Major Origin of Life Theories
To get a clearer picture, it helps to see these leading ideas side-by-side. Each theory offers a different perspective on the location, energy source, and the very first crucial steps toward life.
| Theory | Proposed Location | Key Mechanism | First Major Molecule |
|---|---|---|---|
| Primordial Soup | Warm, shallow ponds or oceans | UV light & lightning on simple gases | Amino Acids |
| Hydrothermal Vent | Deep-sea alkaline vents | Chemical & heat gradients | Simple metabolic cycles |
| RNA World | Unspecified (could fit with others) | RNA self-replication & catalysis | RNA (Ribozymes) |
| Lipid World | Unspecified (could fit with others) | Self-assembling lipid bubbles | Protocells (Vesicles) |
This table isn’t about picking a winner. Instead, it shows how different theories tackle different parts of the problem—some focus on the setting, some on the genetic code, and others on the container.
The Lipid World Hypothesis
While RNA and proteins are the stars of the show, the Lipid World Hypothesis argues that we’re forgetting the stage itself: the container. This theory suggests the first critical step toward life wasn’t a complex molecule, but a simple, self-assembling bubble made of lipids (fatty molecules).
On early Earth, these lipid molecules would have naturally formed enclosed spheres called vesicles when in water. This process spontaneously creates a basic cell membrane, separating an “inside” from an “outside.”
These primitive “protocells” could trap other molecules floating around in the soup. By concentrating these chemicals, they created tiny, isolated labs where the reactions needed for life had a much better shot at happening. This “container-first” model proposes that a basic metabolism could have fired up inside these lipid bubbles long before any complex genetic system like RNA ever came into being.
Understanding the Immense Chemical Diversity of Early Earth

The term “soup” might make you think of something simple, but the chemical reality of early Earth was anything but. We’re talking about a chemical universe teeming with possibilities, far more complex than a simple broth. This wasn’t just a few molecules bumping into each other; it was a dynamic system with staggering combinatorial power.
The latest research really drives this point home. It turns out that even a small, limited set of starting ingredients can explode into an incredible variety of products, highlighting the raw creative force of the primordial soup.
A Mind-Boggling Chemical Universe
To get a real sense of this scale, look at what happens in the lab. Scientists took just a few types of amino acids and hydroxy acids—both believed to be common on early Earth—and watched what happened. Even with these tight restrictions, the results were astonishing.
In one experiment using only three types of amino acids and three types of hydroxy acids, researchers ended up with over 650 different protein precursors known as depsipeptides. When you scale that up, the numbers get truly wild. If the experiment had used 10 amino acids and 10 hydroxy acids, the number of unique molecules could have ballooned to over 10 trillion. You can read the full research on these chemical possibilities.
This illustrates a fundamental point about the primordial soup: its strength wasn’t just in making a few key molecules, but in generating a colossal library of chemical structures. Nature could then select the most stable and useful ones from this enormous pool of options.
The Power of Wet-Dry Cycles
So how did these simple building blocks manage to string themselves together into longer, more useful chains? A giant ocean would have just diluted everything, making it almost impossible for molecules to meet and connect. The key, it seems, was the natural rhythm of the environment.
Picture the shores of ancient tidal pools or the edges of volcanic hot springs. These places would have been locked in a constant cycle of getting wet, then drying out.
- Wet Phase: During high tide or a downpour, water would collect, dissolving simple molecules like amino acids and letting them mix freely.
- Dry Phase: As the sun baked the landscape and the water evaporated, those dissolved molecules would become incredibly concentrated.
This constant cycling would have been the engine for polymerization, forcing the packed-in monomers to link together into longer chains. It’s a beautifully simple, repetitive process that offers a plausible way to bridge the gap from random chemistry to the organized structures of life. This is where prebiotic chemistry and computational biology really start to overlap; if you’re curious about how data science helps us model these processes, you can learn more about what is bioinformatics in our article.
Common Questions About the Primordial Soup Theory
As we’ve journeyed through the science and history of the primordial soup, a few big questions tend to pop up. Let’s tackle some of the most common ones head-on.
Did the Miller-Urey Experiment Actually Create Life?
No, it didn’t—and it was never meant to. The experiment’s real triumph was showing that organic molecules, the very stuff of life, could form on their own.
By zapping a mix of simple chemicals thought to exist on the early Earth, Stanley Miller and Harold Urey created amino acids. They proved that life’s basic building blocks were likely just lying around, waiting for the next step. They didn’t make a cell, but they showed how the raw materials could have appeared naturally.
Is the Primordial Soup Theory Still Relevant Today?
Absolutely. While the original, planet-wide “warm little pond” idea has been refined, the core concept is still the foundation of origin-of-life research. The central idea—that life emerged from non-living chemistry—hasn’t gone anywhere.
Think of it this way: modern theories about deep-sea hydrothermal vents or bubbling volcanic pools aren’t replacements for the primordial soup. They’re just proposing more specific, concentrated “bowls” of soup where the chemistry could happen more efficiently.
What Is the Biggest Challenge for the Primordial Soup Theory?
The single biggest hurdle is often called the “concentration problem.” Imagine a few useful molecules forming in a planet-sized ocean. The chances of them ever bumping into each other to create something more complex would be astronomically low. They’d just be too diluted.
To get around this, scientists now focus on smaller, contained environments where these chemicals could have been concentrated. A few leading ideas include:
- Tidal pools: Where constant cycles of wetting and drying would have left behind a thicker, more reactive chemical sludge.
- Clay surfaces: Microscopic layers of clay could have acted like a template, trapping molecules and organizing them into patterns.
- Hydrothermal vents: These deep-sea chimneys provide both the chemical ingredients and the energy gradients needed to fuel reactions in a confined space.
Solving the concentration problem makes the leap from simple chemistry to the building blocks of biology far more plausible. In a similar way, understanding these fundamental building blocks has led to incredible modern advancements. To see how this foundational science inspires new fields, take a look at our guide on what stem cell therapy is.
Discover more from Maxi Journal
Subscribe to get the latest posts sent to your email.