Your phone is on 18%. You glance at the battery icon, lower the screen brightness, and hope it lasts through the commute. Or maybe your laptop used to make it through a full workday and now starts begging for a charger by mid-afternoon. If you drive an EV, you’ve probably watched charging speeds surge at first and then slow down near the top.
All of those everyday moments come from the same hidden process. Tiny charged particles move back and forth inside a sealed cell, while electrons take a different route through your device and do the useful work.
Lithium-ion batteries became the default for modern electronics for a reason. They were commercialized by Sony in 1991, captured 42% of the global rechargeable battery market by 2022, and powered 95% of electric vehicles sold worldwide in 2023, according to the U.S. Department of Energy’s overview of how lithium-ion batteries work. They also offer 100-265 Wh/kg, compared with 30-50 Wh/kg for lead-acid batteries in that same source. That advantage helps explain why the same basic battery idea now powers phones, laptops, cordless tools, home storage systems, and EVs that also connect to bigger conversations about renewable energy systems.
The chemistry sounds abstract until you connect it to real life. Charging speed, battery aging, cold-weather performance, and safety all trace back to what’s happening inside the cell. Once you see that microscopic story clearly, battery behavior stops feeling mysterious.
The Unseen Engine of Modern Life
A lithium battery rarely gets your attention when it’s working well. You notice the app, the camera, the motor, the screen. The battery stays in the background, making all of it possible.
That invisibility is part of what makes batteries so interesting. A phone feels simple from the outside, but inside it sits a compact energy storage system that has to hold a large amount of energy for its size, release it on demand, recharge hundreds of times, and stay safe while doing it.
Why this technology won
Lithium-ion batteries didn’t become common by accident. They fit modern devices because they pack a lot of energy into relatively little mass and volume. In practical terms, that means lighter laptops, slimmer phones, and electric cars with usable driving range instead of golf-cart limitations.
Their rise also changed how people expect products to behave. We now assume that a wireless device should be portable, rechargeable, and powerful. That expectation exists because lithium-ion chemistry made it normal.
Batteries shape modern design as much as processors and screens do. Without compact rechargeable cells, many everyday devices would be bulkier, weaker, or tethered to a wall.
The everyday clues batteries leave behind
You already know more battery science than you think, because you’ve seen the clues:
- Fast charging slows near the top: That isn’t a broken charger. It reflects how the battery is controlled.
- Cold weather hurts performance: The chemistry still works, but the ion movement becomes less cooperative.
- Old batteries feel “smaller”: The battery hasn’t shrunk. It has lost usable capacity and gained internal resistance.
- One weak battery pack ruins a tool or device: In multi-cell packs, the whole group depends on its weakest members.
The useful question isn’t just how do lithium batteries work. It’s why their inner chemistry shows up in such familiar ways. To answer that, you need to start with the parts inside a single cell.
Inside the Battery Anatomy of a Cell
A lithium-ion battery cell has four essential parts: cathode, anode, electrolyte, and separator. If you want a simple mental model, think of two storage rooms connected by a hallway.
One room is the anode. The other is the cathode. The hallway is the electrolyte, which lets lithium ions travel between the rooms. The separator is a thin porous barrier in the middle. It allows ions through, but keeps the two sides from touching directly and causing a short circuit.
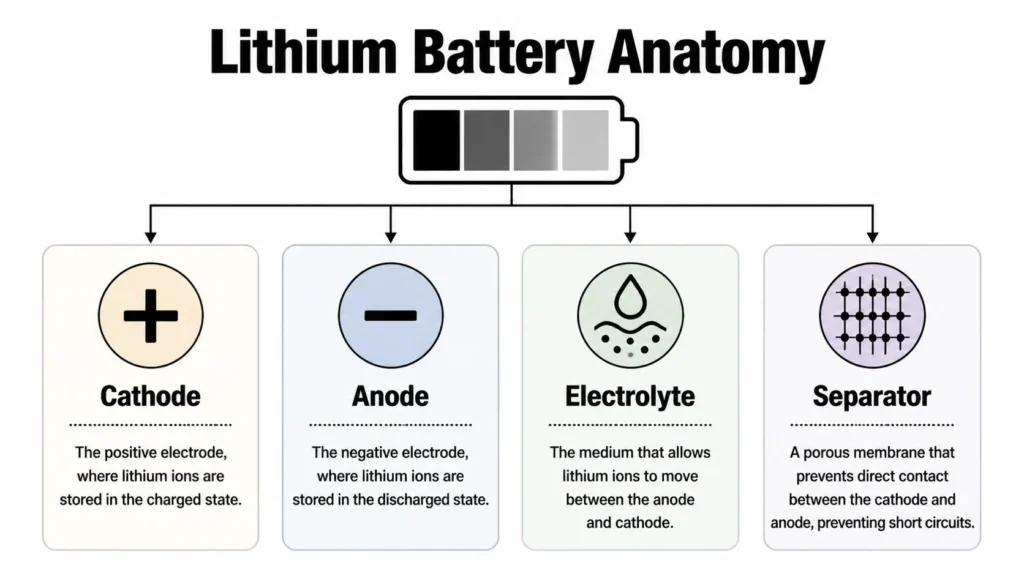
The four parts and their jobs
Here’s the anatomy in plain language:
- Cathode: This is the positive electrode. It contains a lithium-bearing metal oxide material and acts as one of the battery’s main lithium hosts.
- Anode: This is the negative electrode, commonly made from graphite. Graphite works well because lithium ions can slip between its layered carbon structure.
- Electrolyte: This is the ion-moving medium. It doesn’t carry electrons the way a wire does. Its job is to let lithium ions move internally.
- Separator: This thin membrane is a safety-critical part. It keeps the electrodes apart while still allowing ion flow.
A lot of confusion starts here because people assume electricity inside the battery works the same way everywhere. It doesn’t. Electrons move through the external circuit. Lithium ions move inside the battery through the electrolyte.
Why lithium is the star
Lithium is unusually well suited for this job. The University of Washington’s Clean Energy Institute notes that lithium’s small atomic weight and radius, third only to hydrogen and helium, help produce high voltage and energy density, and its single outer-shell electron makes it highly reactive and ready to ionize in a battery system, as explained in their page on the lithium-ion battery.
That combination matters. A battery works by moving charge back and forth in a controlled way. Lithium is light, small, and willing to give up its outer electron. Those traits make it excellent for storing a lot of energy without making the battery excessively heavy.
Practical picture: Lithium is like a very small traveler carrying an easy-to-surrender ticket. Because it’s tiny, it can fit into material structures that would resist larger ions. Because it ionizes readily, it helps create the electrical separation the battery needs.
Why graphite works so well
Graphite deserves more credit than it gets. Its carbon atoms form strong layers, and lithium ions can insert between those layers through intercalation without badly warping the structure. That’s a big reason rechargeable lithium-ion cells can survive repeated cycling instead of falling apart after a few uses.
Once you understand the layout, the next step is motion. A battery isn’t useful because of what it contains. It’s useful because of what moves.
The Electric Dance of Ions
Your phone can drop from 20% to dead shockingly fast on a freezing morning, then seem to regain some life once it warms up. That odd behavior starts with a tiny traffic pattern inside the battery. Charge only flows because two kinds of particles take two different routes.
When the battery is powering a device, lithium ions move inside the cell while electrons move through the outside circuit. The ions travel through the electrolyte. The electrons travel through your phone, laptop, flashlight, or car electronics. That split path is what turns stored chemical energy into usable electric current.

What happens during discharge
During discharge, the battery is doing the job you bought it for.
At the anode, lithium atoms give up electrons and become Li+ ions. The separator blocks electrons, so they cannot cut straight across the battery. They are forced to take the long route through the external circuit. As they pass through that circuit, they power the device.
Meanwhile, the Li+ ions move through the electrolyte toward the cathode, where they reunite with electrons. A simple way to picture it is a two-lane system. One lane is inside the battery for ions. The other lane is outside the battery for electrons. The useful work happens because those lanes stay separate until the particles meet again.
That separation explains a lot of daily battery behavior. If ions can move easily, the battery can supply power quickly. If their movement is slowed, the device feels weak even when some charge remains.
What happens during charging
Charging runs the same system in reverse. An external power source pushes electrons back toward the anode and drives lithium ions back through the electrolyte to be stored there again. The charger is doing chemical lifting, forcing the battery uphill into a higher-energy state that it can use later.
In graphite-based anodes, lithium slips between layers of carbon rather than permanently bonding into a new solid compound. Materials scientists often describe this with the formula LiC6, which reflects the fully lithiated graphite structure, as explained in a Battery University overview of lithium-ion chemistry. That detail helps explain both why graphite is common and why charging speed has limits. Those layers can only accept lithium so fast before side reactions and heat become a problem.
The same basic idea shows up in many modern technologies that rely on controlled prediction and monitoring, from battery charging algorithms to machine learning systems that learn from patterns in data.
Why this movement creates usable energy
A battery works because the materials on each side prefer different chemical states. Engineers choose those materials so electrons have a reason to leave one side and travel to the other through a circuit. Your device taps into that journey.
So the battery is less like a bucket of electricity and more like a spring held under tension. Charging tightens the spring by separating charges into less stable positions. Discharging lets the system relax in a controlled way, and that relaxation becomes electrical power.
Why cold weather and fast charging feel different
Cold changes the pace of the dance. Lithium ions move more slowly through the electrolyte, and they also enter electrode materials less easily. Internal resistance rises. Voltage sags more under load. Your phone may shut off early because the battery struggles to deliver energy fast enough, not because every bit of stored energy is gone.
Fast charging pushes the opposite problem. The charger tries to move lithium ions back quickly, but the ions and electrode surfaces can only keep up to a point. Push too hard, especially at low temperatures, and unwanted reactions become more likely. That is one reason charging speed often slows near full charge and why battery care systems pay close attention to temperature.
Everyday battery life is the visible result of these invisible particle movements. Charging speed, winter performance, and long-term wear all begin here.
The Brains of the Battery Management System
You plug in your phone at 15%, check it again half an hour later, and it has jumped upward. Then the last stretch to full seems to crawl. That familiar pattern is not just chemistry. It is chemistry being supervised.
The supervisor is the Battery Management System, or BMS. In a battery pack, the BMS works like a control room. It watches what each cell is doing, compares those readings against safe limits, and decides when to allow full power, when to slow charging, and when to shut things down before damage starts.

What the BMS actually does
A lithium cell can store a lot of energy in a small space, but it has rules. Push the voltage too high, drain it too low, or let it get too hot, and side reactions become more likely. Those side reactions are part of the reason batteries age, lose capacity, or in severe cases become unsafe.
The BMS helps prevent that by handling several jobs at once:
- Stops overcharging: It cuts back or stops charging before cells exceed their safe voltage range.
- Stops over-discharging: It prevents the kind of deep drain that can permanently harm the electrodes.
- Tracks temperature: It watches for heat during charging, heavy use, or fast discharge.
- Manages cell differences: In a pack, no two cells age in exactly the same way.
- Talks to the rest of the device: Chargers, phones, laptops, EVs, and power tools all adjust behavior based on battery feedback.
That last point matters in everyday life. The percentage on your screen is not a direct window into the chemistry. It is an estimate produced by electronics and software using voltage, current, temperature, and past behavior.
Why charging starts fast and ends slow
The charging pattern has a name: CC-CV, short for constant current, constant voltage.
Early in the charge, the system usually feeds the battery a steady current. Once the cell reaches its upper voltage limit, the charger switches strategies and holds that voltage steady while the current gradually falls. Battery University explains this standard method in its overview of lithium-ion charging stages and charge termination.
That slowdown near full charge is easy to misread. It can feel like the charger suddenly got worse. What is happening is more like filling seats in a theater. At the beginning, there is plenty of room and people can move in quickly. Near the end, the remaining open spots are harder to reach without bumping into the safety limits that protect the cell.
So the BMS eases off on purpose.
Charging rule: The slow final stretch is a protection step that reduces stress on the cell.
That control logic is one reason battery systems are often discussed alongside software-heavy fields like machine learning. The battery is not doing machine learning in this basic process, but modern devices increasingly use prediction and control software to estimate range, charging time, and battery health.
Why balancing matters in packs
A single cell is simple. A pack is a team.
In a phone, laptop, e-bike, or EV pack, small differences between cells build up over time. One cell may fill a little faster. Another may warm up more under load. Another may age sooner. If the pack ignored those differences, the weakest or fullest cell would reach its limit first and set the boundary for the whole pack.
The BMS handles that mismatch with cell balancing. During charging, it can trim back the cells that are getting ahead so the rest can catch up. The goal is not perfection. The goal is to keep the pack working as one coordinated system instead of a tug-of-war between slightly different cells.
This helps explain a real-world mystery. Two battery packs with the same chemistry can age differently because their cells, cooling, charging habits, and control systems are different. The chemistry stores the energy. The BMS shapes how gently or harshly that chemistry is used over months and years.
A quick visual helps if you want to see pack electronics in context.
A Family of Power Chemistries and Forms
You see the difference every day, even if the battery is hidden.
One phone lasts all day but charges a little slower. One EV keeps more range after years of use. A home backup battery is heavier than a laptop battery, yet people trust it to sit in a garage and cycle for years. Those differences often come from chemistry choice and cell shape, not just battery size.
“Lithium battery” is really a family name. The cells all move lithium ions between two electrodes, but they use different cathode materials, and those materials change the battery’s personality. Some chemistries are calmer under stress and hold up well over many cycles. Others store more energy in the same space, which is why they show up in thinner or longer-range devices.
Common lithium-ion chemistries compared
| Chemistry (Abbreviation) | Primary Advantage | Main Trade-off | Typical Application |
|---|---|---|---|
| LFP | Strong safety profile and durability | Lower energy density than some alternatives | Grid storage, many EVs, backup power systems |
| NMC | Good balance of energy and performance | More complex trade-offs around cost, thermal behavior, and materials | EVs, e-bikes, consumer electronics |
| NCA | High energy density | Narrower safety margin than more stable chemistries | Some EV packs and high-performance applications |
| LiCoO2 | Well suited to compact electronics | Trade-offs in cost, thermal behavior, and lifespan depending on design | Phones, laptops, small electronics |
LFP is a good example of how these trade-offs show up in real products. The U.S. Department of Energy explains that lithium iron phosphate uses strong phosphate bonds, which help make the chemistry more thermally stable, while its lower cell voltage and energy density can make packs larger for the same stored energy in many designs. That is one reason LFP often appears in stationary storage and in EVs built around long life and safety rather than maximum range. See the DOE overview of lithium iron phosphate batteries.
Chemistry choice is an engineering decision
A phone designer usually wants lots of energy in a very small, light package. A grid storage engineer often cares more about long cycle life, predictable behavior, and lower cost per cycle. EV makers sit in the middle and choose based on range targets, charging performance, pack size, and price.
That is why there is no single “best” lithium-ion battery.
Chemistry also helps explain a few everyday mysteries. A battery built for high energy density may give a slim device longer runtime at first, but it can be less forgiving of heat and stress. A chemistry chosen for durability may charge and discharge more steadily over years, even if it needs more space. The microscopic structure of the cathode ends up shaping things people notice at the wall outlet, on the dashboard, and in winter weather.
Shape matters too
The same chemistry can be packaged in different forms, and the shape changes how engineers cool, protect, and assemble the pack.
- Cylindrical cells: Common in power tools and some EV packs. They are mechanically strong and consistent in mass production.
- Pouch cells: Lightweight and space-efficient, often used where designers want thin or flexible layouts.
- Prismatic cells: Box-shaped cells that can make pack layout simpler in larger devices.
Form factor affects more than appearance. It influences how tightly cells can be packed, how easily heat can escape, and how much structural support the product needs around the cell. In a phone, those choices affect thickness and internal layout. In an EV or factory-built battery pack, they connect directly to automated assembly, sensors, and robotic manufacturing systems.
Chemistry sets the cell’s tendencies. Form decides how those tendencies fit into a real machine.
Why All Batteries Eventually Die
You notice it on an ordinary day. Your phone reaches 20 percent by late afternoon when it used to cruise past dinner. Nothing about the outside looks different, but inside the cell, the chemistry has been slowly changing since the day the battery was first used.
A lithium-ion battery ages because charging and discharging are not perfectly reversible. The cell does not move lithium ions back and forth forever without side effects. Small unwanted reactions happen alongside the useful ones, and those side reactions gradually take away lithium the battery can still use.
The slow buildup inside the cell
One of the first ideas to get clear is the SEI, or solid electrolyte interphase. It forms on the anode surface as the battery begins operating. In moderation, that layer helps. It acts like a thin protective skin between the anode and the electrolyte, reducing further direct reaction.
The problem is that the skin does not stay frozen in time.
As battery researchers at the Battery University explanation of lithium-ion aging describe, side reactions continue during use and storage, especially under stress. As the SEI grows thicker, it consumes active lithium and increases internal resistance. That means the battery has less charge to work with, and it also has a harder time delivering power quickly.
This helps explain a familiar experience. An older phone may still show a decent percentage, yet it can shut down sooner during a demanding task like video recording, gaming, or using GPS in the cold. Part of the issue is lost capacity. Part is rising resistance inside the cell, which makes voltage sag more under load.
SEI buildup works like mineral scale inside a pipe. The system still functions, but flow becomes harder.
Why heat, full charge, and fast charging age batteries faster
Not all battery time is equally stressful. Heat speeds up chemical reactions, including the unwanted ones. Holding a battery near full charge also pushes the materials into a more chemically strained state. Fast charging can add stress because it moves ions quickly and generates more heat, especially if the battery is already warm.
That is why two identical devices can age at different rates. One spends hours in a hot car, stays at 100 percent overnight in a warm room, or fast-charges often during heavy use. The other lives a calmer life. The difference shows up months later as shorter runtime, slower charging near the top of the battery meter, or sudden drops in percentage.
Cold creates a different problem. It usually does not do as much long-term damage as heat, but it slows ion movement inside the battery. The battery then struggles to deliver current, so your phone can feel weak in winter even if its long-term health has not changed much. This is the microscopic chemistry showing up as an everyday annoyance.
The weakest cell problem
In a multi-cell battery pack, aging is also uneven. Cells are made to tight standards, but no two are perfectly identical. Over time, tiny differences in capacity and resistance grow larger. One cell may fill sooner, empty sooner, or heat up more than its neighbors.
The pack has to respect that weakest cell. If one cell reaches its safe voltage limit early, charging or discharging for the whole pack must be adjusted around it. That is one reason a laptop battery, e-bike pack, or EV module can feel worn out even though several cells inside still have useful life left.
Wikipedia’s overview of lithium-ion batteries notes that multi-cell packs need balancing because cell mismatch grows over time. In practice, that means battery aging is not just a story about one cell losing capacity. It is also a story about a group losing coordination.
A battery pack ages more like a team than a single block. One struggling cell can lower the performance of the whole group.
Using Your Batteries Safely and Wisely
Most battery advice sounds like folklore until you understand the chemistry behind it. Then the rules make sense.
If a battery gets too hot, becomes physically damaged, or is charged outside safe limits, reactions inside the cell can become self-accelerating. That’s the danger behind thermal runaway. In plain language, the battery starts making heat faster than it can shed heat, and the situation can escalate.

Habits that help
You don’t need a lab to treat batteries better. A few habits go a long way:
- Keep them out of extreme heat: Don’t leave a phone baking on a dashboard or a laptop under blankets while charging.
- Use the intended charger and cable: The device and charger are designed to work with the battery’s control system.
- Avoid physical abuse: Crushing, puncturing, or bending a battery can damage internal layers.
- Watch for swelling: A swollen phone, trackpad, or battery pack is a warning sign, not a cosmetic issue.
- Charge thoughtfully when possible: Many devices manage this well for you, but avoiding long periods at stressful conditions can help.
Why cold changes performance but heat does more damage
Cold mostly makes batteries sluggish. Heat is more destructive.
In the cold, ion movement slows and power delivery drops. In heat, unwanted chemical reactions speed up. So the battery may feel weak in winter, but it often ages faster during hot summers, fast charging in warm conditions, or heavy use with poor cooling.
What safety features are already protecting you
Modern devices don’t leave safety to luck. They often include:
- Voltage limits enforced by charging electronics
- Temperature monitoring during use and charging
- Current limits to reduce stress
- Pack shutdown behavior when the battery enters unsafe conditions
- Cell balancing in larger packs
Those protections are why millions of people use lithium-ion batteries every day with little thought. Still, electronics can’t fix everything. A damaged or swollen battery should be replaced, and a pack that behaves strangely deserves caution.
Don’t ignore a battery that gets unusually hot, swells, leaks, or smells odd. Those are signs to stop using the device and handle it carefully.
The nice part of learning how do lithium batteries work is that it turns random advice into understandable cause and effect. Fast charging slows near full because the battery needs protection. Old packs fade because internal resistance grows and usable lithium gets tied up. Cold weather cuts performance because ion motion slows down. The science is small-scale, but the effects are right there in your hand every day.
If you enjoy clear explanations of science and technology without the jargon overload, visit maxijournal.com. You’ll find approachable articles across tech, science, health, business, arts, travel, and more, along with fresh writing for curious readers who like understanding how things work.
Discover more from Maxi Journal
Subscribe to get the latest posts sent to your email.


